Researchers have literally unearthed clues as to why the 1918 influenza pandemic was so deadly. The 1918 influenza pandemic ranks as the largest and most destructive outbreak of an infectious disease, killing 20 to 40 million people worldwide. Using fragments of the flu genome from Alaskan victims preserved by permafrost and army autopsy tissues, James Stevens and Ian Wilson of the Scripps Research Institute in La Jolla, California and their collaborators have assembled genes from the 1918 flu virus.
Approximately 1,700 scientists visit SSRL annually to conduct experiments in broad disciplines including life sciences, materials, environmental science, and accelerator physics. Science highlights featured here and in our monthly newsletter, Headlines, increase the visibility of user science as well as the important contribution of SSRL in facilitating basic and applied scientific research. Many of these scientific highlights have been included in reports to funding agencies and have been picked up by other media. Users are strongly encouraged to contact us when exciting results are about to be published. We can work with users and the SLAC Office of Communication to develop the story and to communicate user research findings to a much broader audience. Visit SSRL Publications for a list of the hundreds of SSRL-related scientific papers published annually. Contact us to add your most recent publications to this collection.
New evidence is overturning the assumption that the thermal oxide grown on single crystal silicon is completely amorphous. SSRL researchers Anneli Munkholm (now at Lumileds Lighting) and Sean Brennan recently published work in Physical Review Letters showing that there is residual order within the oxide. The thermal oxide film on silicon has been studied extensively for decades because these oxides form the basis of most of the integrated circuits used in modern electronics. The new results have implications for theoretical models of the oxidation process.
Extremely small pieces of a material aren't always a chip off a bigger block. How nanomaterials behave is tremendously important to know when trying to understand the roles of mineral nanoparticles in the environment, or design devices for nanotechnology. Researchers taking data at SSRL and the Advanced Photon Source (APS) in Illinois recently found that zinc sulfide at 3.5 nanometers (nm) in size (3.5 billionths of a meter) behaves quite differently than "bulk" zinc sulfide (several hundred nm and up). The method they developed should also prove useful for studying other kinds of nanomaterials.
Anthrax makes a deadly cocktail of three toxin proteins that flood the bloodstream, leading to rapid death if the infection is not diagnosed and treated in its early stages. Even antibiotic treatments can fail when the Anthrax bacterium, Bacillus anthracis, has already produced lethal levels of toxins. The poisonous protein called Lethal Factor (LF) rapidly blocks signals to recruit immune cells to fight the infection. Another enzyme Edema Factor (EF) causes the release of fluid into the lungs and is deadly on its own.
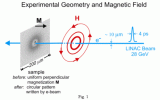
Two important goals of technology are: smaller and faster. In line with this goal the 50 billion dollar per year magnetic recording industry has long wondered where it would run into obstacles set by fundamental physical principles. The basic questions concern the lateral sizes at which the magnetic domains that define the "1" and "0" bits become unstable and whether there is a speed limit for the writing process of the bits.
Antibiotics and the bacteria they attack are engaged in a constant race to out-evolve one another. An antibiotic is effective against specific bacteria only so long before the random mutations that all bacteria undergo make them resistant to that particular drug. Recently, scientists from the University of Georgia, Utah State University, and Guilford Pharmaceuticals carried out studies at SSRL that could enable drug designers to pull ahead, at least for a while, by developing a new class of antibiotics.
In ice, each water molecule is surrounding by 4 other molecules in a tetrahedral arrangement (left). The new result on liquid water shows that the molecules are connected only with 2 others. This implies that most molecules are arranged in strongly hydrogen bonded rings (middle) or chains (right) embedded in a disordered clusternetwork connected mainly by weak hydrogen bonds. The oxygen atoms are red and the hydrogen atoms grey in the water (H2O) molecules.
Toxic and carcinogenic chromate (hexavalent chromium as CrO42-) has contaminated the groundwater in Hanford, Washington. At Hanford, hexavalent chromium was used in the industrial process to recover plutonium from irradiated nuclear fuels. The resulting high-level waste corroded its storage tanks and leaked into the desert subsoils. Plumes of contaminated groundwater reaching the Columbia River pose a risk to spawning salmon.
Professor Roger D. Kornberg and his group in the Stanford University School of Medicine have devoted more than 20 years to the study of the process by which genetic information encoded in all living things by DNA is processed into a message (a process called transcription that produces messenger RNA) that then directs the synthesis of proteins. A breakthrough paper detailing the structures of the core RNA Polymerase II protein was published in Science in April 2000 and followed by two more papers in Science a year later.
Computer simulations have shown that by using a cleverly placed piece of slotted foil, the Linac Coherent Light Source (LCLS) will be able to produce brilliant x-ray pulses that are extremely short, a few femtoseconds (a femtosecond is a quadrillionth of a second), in duration. This pulse length, which is a factor of more than 200 times shorter than the LCLS baseline design, will dramatically increase LCLS' x-ray time resolution, giving scientists the ability to study the movement of matter at atomic scales and accessing the structural changes occurring in the making or breaking of chemical bonds.