Chromate (hexavalent chromium as CrO42-) is a significant groundwater contaminant at the U.S. Department of Energy (DOE) Hanford Site in southeastern WA (Poston et al., 2001) where Pu was produced during WWII and the cold war, and where DOE's largest inventory of legacy wastes remain. Chromate in ground water is readily transported through the Pleistocene-age flood deposits at Hanford, resulting in groundwater contamination plumes that discharge CrO42- to the Columbia River, posing risk to spawning salmon and downstream potable water supplies. The present work suggests that ca 42% of the Cr known to be present in the plumes is immobile due to precipitation of relatively insoluble Cr(III) phases, abating somewhat the extent of the problem. The balance of chromium in the plumes occurs as mobile chromate. Defining the chemical and physical forms of the contaminant, such as is presented here, is the crucial first step to mitigating a contaminant problem.

Waste Management Area (WMA) S-SX, the focus of this study, is one of 12 high-level waste tank farms at Hanford. The SX tank farm contains 15 massive, single-shell, high-level waste tanks, each with 3,785 kL (1,000 kgal) capacity. The SX waste tanks received self-boiling waste solutions from Hanford's Reduction-Oxidation (REDOX) Plant in the mid 1950s. The REDOX process was used to recover Pu from irradiated nuclear fuels. Hexavalent Cr [Cr(VI)] in the form potassium dichromate was used as an oxidant in the REDOX process to manipulate the valence states of Pu and U. The REDOX wastes were self-boiling, and self-concentrated from the radioactive decay of short-lived isotopes.
During the 1960s and 1970s nine of the SX tanks developed leaks as a result of the high thermal load from waste boiling and the caustic nature of the wastes. One of the largest leaks was from SX-108 that discharged 57,760 L of hot, highly concentrated REDOX waste to the vadose zone (i.e., the area of the subsurface above the water table) in 1969. The leaked SX-108 tank waste (Table 1) contained high concentrations of salt, base, Cr, 137Cs+,99Tc(VII), and other radioactive and chemical constituents. Hanford personnel collected subsurface samples beneath tank SX-108 in 2000 using a sophisticated slant drilling device to assess the depth distribution and fate of tank waste contaminants. Some of the retrieved core samples were the most radioactive geomedia ever brought to the surface at Hanford.
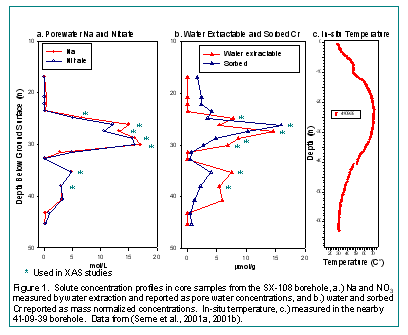
Characterization measurements of the core samples indicated an unusual pattern of chemical attenuation of CrO42- that required explanation for risk assessment (Fig. 1). A well-defined vadose zone plume of high concentration Na-NO3 (Fig. 1a) that was almost coincident with Cr (Fig. 1b) was observed between depths of 23-32 m and 34-42 m in a region of high residual heat (Fig. 1c). Model calculations indicate that temperatures at 25 m approached 100o C at the time of leakage in 1969. The Na-NO3 was freely soluble in water, but in contrast, sizable fractions of the total Cr were sorbed (i.e. associated with solid sediments) and only extractable in strong acid. The limited solubility of sorbed Cr, which was higher in the upper regions of the core, implied that it existed in a precipitated or strongly adsorbed state. Because there was little or no previous evidence for adsorptive retardation of Cr(VI) in oxic Hanford vadose zone sediments, XAS measurements were performed at SSRL to define Cr valence in sediment as a basis for establishing a conceptual model of geochemical reaction.
Cr K-XANES spectra were measured on eight SX-108 samples (asterisk in Fig. 1) using the Molecular Environmental Science Beam Line (BL 11-2) (Bargar et al., 2001) or the general XAFS beamline (BL 4-3) over the energy range 5900 eV to 6350 eV. The samples were extremely radioactive and contained up to 0.1 mCi/g of 137Cs+ (Liu et al., 2003). The methods of Peterson et al. (1997) were used to analyze the Cr(VI) content of the sediments from the intensity of the pre-edge feature. The percent Cr(VI) was calculated from the sample specific pre-edge peak height and positions using the following beamline-specific calibration curves:
BL 11-2: %Cr(VI) = 102.83(height) - 12.80
BL 4-3: %Cr(VI) = 128.64(height) - 12.77
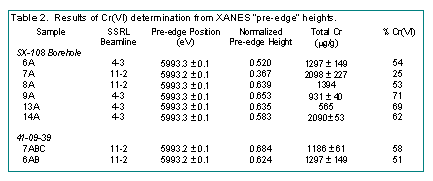
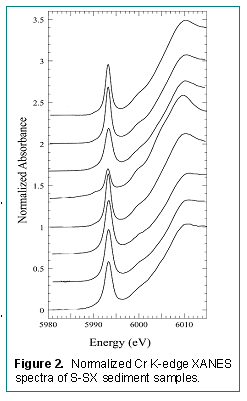
The calibration curve correctly predicted the Cr(VI) percentages of three blind samples that were used to test the calibration process. The normalized Cr K-XANES spectra of the eight Hanford borehole samples all displayed the presence of both Cr(VI) and Cr(III) (Fig. 2, Table 2). The largest Cr(III) concentration [smallest Cr(VI)] was observed in SX-108 Sample 7A, which had the highest pH and significant mineral alteration resulting from reaction of the high pH waste with the sediment. In contrast, the highest Cr(VI) concentrations were noted deep in the core where pH was ambient and where mineral alteration was minimal. These data indicated that Cr(VI) present initially in the REDOX waste was variably reduced by the Hanford sediments, and that reduction extent correlated with sediment mineral alteration.
The XANES measurements and others not described here (Zachara et al., 2004) allowed us to conclude that ferrous iron (Fe2+) was released from the oxic Hanford sediment by base-induced, heat facilitated dissolution of Fe(II)-containing minerals including biotite (iron-bearing aluminuosilicate clay mineral), clinoclore (chlorite aluminosilicate), and ilmenite (FeTiO3). The Fe(II) so liberated was an effective reductant of Cr(VI), which then precipitated as an insoluble Cr(OH)3 phase or was incorporated within iron-bearing precipitated phases. This reductive precipitation reaction apparently occurred after the waste plume had reached its current configuration and depth. The results demonstrate that a minimum of 42% of the total Cr inventory is effectively immobilized as Cr(III) precipitates that are unlikely to dissolve appreciably under the low drainage conditions of the Hanford vadose zone. The remaining Cr(VI), however, will be free to migrate to groundwater unless surface infiltration at the tank farm surface is controlled. These and other ongoing studies at SSRL on contaminated sediments from different Hanford tank farms are providing key scientific insights on the hazards posed by such extreme chemical and radioactive materials and the most effective, long-term environmental management strategies to deal with them.
- Poston T. M., R. W. Hanf, R. L. Dirkes, and L. F. Morasch. 2001. Hanford Site Environmental Report for Calendar Year 2000. Pacific Northwest National Laboratory, Richland, WA.
- Jones, T. E., R. A. Watrous, and G. T. Maclean. 2000. Inventory Estimates for Single-Shell Tank Leaks in S, and SX Tank Farms, RPP-6285, CH2M HILL Hanford Group, Inc., Richland, Washington.
- Serne R. J., H. T. Schaef, B. N. Bjornstad, B. A. Williams, D. C. Lanigan, D. G. Horton, R. E. Clayton, V. L. LeGore, M. J. O'Hara, C. F. Brown, K. E. Parker, I. V. Kutnyakov, J. N. Serne, A. V. Mitroshkov, G. V. Last, S. C. Smith, C. W. Lindenmeier, J. M. Zachara, and D. B. Burke. 2001a. Geologic and Geochemical Data Collected From Vadose Zone Sediments from Borehole SX 41-09-39 in the S/SX Waste Management Area and Preliminary Interpretations. Pacific Northwest National Laboratory, Richland, WA.
- Serne R. J., G. V. Last, G. W. Gee, H. T. Schaef, D. C. Lanigan, C. W. Lindenmeier, R. E. Clayton, V. L. LeGore, R. D. Orr, M. J. O'Hara, C. F. Brown, D. B. Burke, A. T. Owen, I. V. Kutnyakov, and T. C. Wilson. 2001b. Geologic and Geochemical Data Collected From Vadose Zone Sediments From Borehole SX 41-09-39 in the S/SX Waste Management Area and Preliminary Interpretations. Pacific Northwest National Laboratory, Richland, WA.
- Bargar, J. R., G. E. Brown, Jr., I. Evans, T. Rabedeau, M. Rowen, and J. Rogers. 2001. A new hard X-ray XAFS spectroscopy facility for environmental samples, including actinides, at the Stanford Synchrotron Radiation Laboratory. Proc. 2nd Euroconference and NEA Workshop on Speciation, Techniques, and Facilities for Radioactive Materials at Synchrotron Light Sources.
- Liu, C., J. M. Zachara, S. C. Smith, J. P. McKinley, and C. C. Ainsworth. 2003. Desorption kinetics of radiocesium from subsurface sediments at Hanford Site, USA. Geochim. Cosmochim. Acta 67(16), 2893-2912.
- Peterson, M. L., A. F. White, G. E. Brown Jr., and G. A. Parks. 1997. Surface passivation of magnetite by reaction with aqueous Cr(VI): XAFS and TEM results. Environ. Sci. Technol. 31, 1573-1576.
- Zachara, J. M., C. C. Ainsworth, G. E. Brown, Jr., J. G. Catalano, J. P. McKinley, O. Qafoku, S. C. Smith, J. E. Szecsody, S. J. Traina, and J. A. Warner. 2004. Chromium speciation and mobility in a high level nuclear waste vadose zone plume. Geochim. Cosmochim. Acta68(1), 13-20.
J. M. Zachara, C. C. Ainsworth, G. E. Brown, Jr., J. G. Catalano, J. P. McKinley, O. Qafoku, S. C. Smith, J. E. Szecsody, S. J. Traina and J. A. Warner, "Chromium Speciation and Mobility in a High Level Nuclear Waste Vadose Zone Plume", Geochim. Cosmochim. Acta 68, 13 (2004)

