The development of broadly neutralizing antibodies (bnAbs) against HIV represents a cornerstone goal of modern vaccine research. VRC01-class bnAbs are of particular interest because they target a conserved site on HIV used to enter human cells and can neutralize diverse viral strains. However, the B cells capable of producing these antibodies are extremely rare and respond poorly to conventional vaccines. One promising strategy to overcome this challenge is germline-targeting vaccination, in which the immunogen is specially designed to seek out and activate precisely these rare B cells. The eOD-GT8 60mer-nanoparticle was engineered by the Schief lab at Scripps with this purpose in mind, and a Phase 1 human clinical trial (IAVI G001) showed that it successfully triggered the desired B cell response in 97% of vaccinated participants [1]. However, how exactly these early antibodies engaged the vaccine antigen at the molecular level, and whether they already resembled mature bnAbs in their binding, was not yet understood.
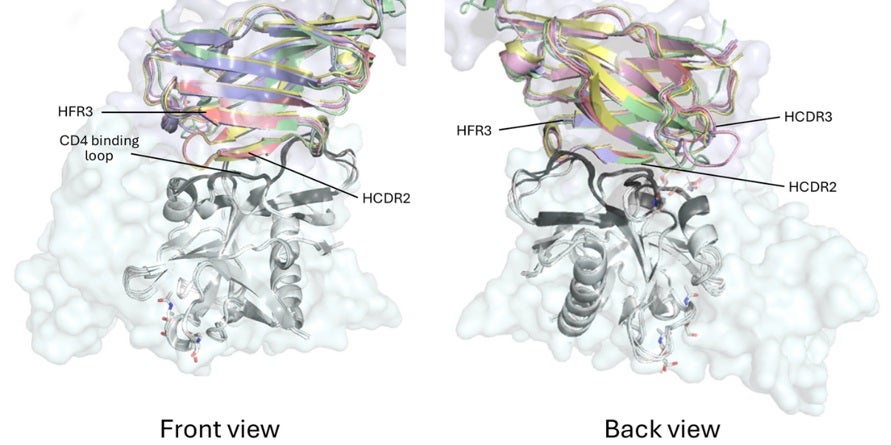
In the current study, Xiaohe Lin in the Wilson lab determined high-resolution X-ray crystal structures of five representative VRC01-class bnAb precursor Fab fragments isolated from trial participants, in complex with eOD-GT8, using SSRL BL12-1 and the APS. Despite being nearly identical to their germline sequences and carrying very few mutations, all five precursor antibodies bound the CD4 binding site (CD4bs) in a manner closely resembling that of mature bnAbs (Fig. 1). The heavy chain drove most of this recognition, making the same key contacts with the antigen as seen in fully mature antibodies. The light chains were more variable, but similar to those elicited by natural infection. Each contributed additional stabilizing contacts, and all five shared a short five-residue loop (LCDR3) that is required to fit within the binding site without steric clash.
The researchers also examined how these early antibodies handle the N276 glycan—a sugar on the surface of HIV that is a known obstacle to bnAb recognition and was not present on the vaccine immunogen. Structures of two precursors bound to a glycan-containing version of eOD-GT8 showed that each used a different strategy to make room for the glycan, without needing the mutations that mature bnAbs rely on. Testing 230 antibodies from trial participants confirmed that 87% could still bind the glycosylated antigen, although with 58-fold reduced affinity, demonstrating that the capacity to accommodate this key glycan is present even at the earliest stages of the immune response to this vaccine immunogen.
In summary, this work provides the first structural evidence that germline-targeting vaccination in humans can activate VRC01-class bnAb precursors capable of mimicking the binding mode of mature bnAbs, despite minimal somatic hypermutation. The findings show that the many of the antibody features required for HIV recognition are already hardwired into our DNA and emerge intact when the right vaccine is given. The capacity for N276 glycan accommodation at the precursor stage, together with the preservation of key germline-encoded interactions at the CD4bs, supports the feasibility of guiding these early intermediates toward broad neutralization through iterative booster immunization strategies.
Primary Citation
X. Lin, C. A. Cottrell, O. Kalyuzhniy, R. Tingle, M. Kubitz, D. Lu, M. Yuan, W. R. Schief and I. A. Wilson, "Structural Insights into VRC01-class bnAb Precursors with Diverse Light Chains Elicited in the IAVI G001 Human Vaccine Trial", Proc. Natl. Acad. Sci. USA 122, e2510163122 (2025) doi: 10.1073/pnas.2510163122
References
David J. Leggat et al. “Vaccination induces HIV broadly neutralizing antibody precursors in humans”, Science 378, eadd6502 (2022). DOI:10.1126/science.add6502