Lithium ion batteries are used ubiquitously for portable energy storage in today’s modern electronic devices and have served in that capacity for decades. Recently, budding energy storage markets — such as those of electric vehicles, large-scale renewable energy storage, and grid balancing — have emerged that require storage capabilities that are beyond what today’s lithium ion technologies currently provide. Specifically, in order for these markets to grow and be cost-effective, they require batteries that can last longer and store more energy per weight, volume, and dollar. Since only 50-60% of the total storage capacity in the widely employed layered transition metal oxide positive electrodes, such as LiCoO2 and LiNi1/3Co1/3Mn1/3O2, is accessed in commercial cells, increasing the accessed capacity by raising the charging cutoff voltage is often considered to be the lowest hanging fruit for increasing energy density. However, this comes at a cost in the form of rapid capacity fade and increased cell impedance.[1] Thus a significant research effort has focused on improving the cycle life of these electrodes during high voltage cycling.
Understanding the mechanisms through which the high voltage degradation occurs is crucial to developing workarounds that tackle the problem. To that end, several capacity fade mechanisms have been identified including atomic-scale transformations[2] and cracking of secondary agglomerate microstructures.[3] Now, through a collaboration between Stanford University and SSRL, Gent et al. have uncovered a surprising new capacity fade mechanism that could lead to novel ways of improving the performance of layered oxide electrodes at high voltage. The discovery reveals the crucial role of the secondary particle agglomerates, which are a commonly used material morphology in layered oxide electrodes due to their ease of synthesis, in capacity fade.
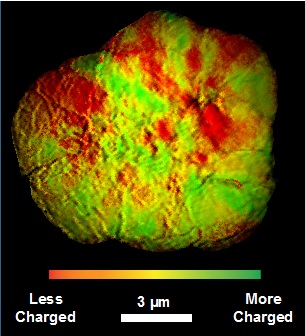
The original goal of the collaboration was to use transmission x-ray microscopy, developed at SSRL, to image the dynamic movement of lithium within LiNi1/3Co1/3Mn1/3O2 secondary agglomerates, which are approximately 10-15 μm large and consist of hundreds of smaller primary nanoparticles. To observe dynamic behavior, the researchers planned to image the particles in situ while they were being actively charged and discharged in a real battery. However, to first confirm whether the technique could adequately resolve the changes in local lithium content – or state-of-charge (SOC) – the researchers compared secondary particles charged to different SOCs and imaged ex situ. Since the particles would wait over 10 h between being actively charged and imaged, it was expected that any internal SOC heterogeneity due to lithium migration would have relaxed and these particles would have a uniform internal distribution of SOC, though with different average SOCs.
Surprisingly, despite the long relaxation time, the particles exhibited strongly non-uniform internal SOC distributions, with regions within individual secondary particles differing in SOC by over 10%. Since layered transition metal oxides exhibit solid solution phase behavior with respect to lithium content,[4] it is typically assumed that at equilibrium they should be entirely uniform. It was already surprising that 10 h appeared to be insufficient to achieve uniformity, so the researchers imaged particles prepared under a variety of different conditions in an attempt to measure how long it took for the particles to fully relax. However, regardless of how slow the particles were charged or how long they were relaxed for, the SOC heterogeneity always persisted to the same degree. The only particles that didn’t exhibit heterogeneity were fully discharged particles. Thus it seemed that the secondary particles were in fact able to fully relax in all cases but they just exhibited SOC heterogeneity at equilibrium – essentially, lithium was getting trapped in certain domains indefinitely.
To explain this, the researchers hypothesized that the secondary particles build up non-uniform stresses during charge that result in a thermodynamic preference for lithium to reside in certain domains over others. The stresses result from the fact that the primary particles are randomly oriented within the secondary agglomerates and anisotropically change shape and volume during charge. Essentially, they don’t change volume cooperatively and the competing forces generate stress. Crucially, since the stresses cannot relax, SOC heterogeneity will always be present in LiNi1/3Co1/3Mn1/3O2 electrodes even at infinitely slow charging rates. Therefore at the charging cutoff voltage there will always be domains that are overcharged and have an SOC greater than the average. Thus even though a cutoff voltage may be chosen to minimize capacity fade based on the average electrode SOC at this voltage, the overcharged domains will have an even greater SOC than the average and will therefore degrade more rapidly than expected. This unusual capacity fade mechanism was previously unknown, yet it provides some rationalization for studies that have shown improved cycle lives for more layered oxide electrodes with more porous secondary structures.[5,6]
Through these observations the authors have highlighted the crucial importance of controlling the secondary particle morphology in tackling high voltage capacity fade in layered transition metal oxide electrodes. Their results also show that it is possible to improve the cycle life of already commercialized materials by improving their morphology – for example, by making them more porous – to minimize the stress buildup and resulting SOC heterogeneity during charge. Moving forward, experimental groups in industry and academia can use this knowledge to rationally design electrode materials with higher energy densities and longer cycle lives.
[1] K. J. Nelson, G. L. d'Eon, A. T. B. Wright, L. Ma, J. Xia, J. R. Dahn, J. Electrochem. Soc. 162, A1046 (2015).
[2] F. Lin, I. M. Markus, D. Nordlund, T.-C. Weng, M. D. Asta, H. L. Xin, M. M. Doeff, Nat. Commun. 5, 3529 (2014).
[3] S. Watanabe, M. Kinoshita, T. Hosokawa, K. Morigaki, K. Nakura, J. Power Sources 260, 50 (2014).
[4] B. J. Hwang, Y. W. Tsai, D. Carlier, G. Ceder, Chem. Mater. 15, 3676 (2003).
[5] K. M. Shaju, P. G. Bruce, Adv. Mater. 18, 2330 (2006).
[6] W.-B. Hua, X.-D. Guo, Z. Zheng, Y.-J. Wang, B.-H. Zhong, B. Fang, J.-Z. Wang, S.-L. Chou, H. Liu, J. Power Sources 275, 200 (2015).
W. E. Gent, Y. Li, S. Ahn, J. Lim, Y. Liu, A. M. Wise, D. N. Mueller, C. B. Gopal, R. Davis, J. Nelson Weker, J.-H. Park, S.-K. Doo and W. C. Chueh, "Persistent State-of-Charge Heterogeneity in Relaxed, Partially Charged Li1−xNi1/3Co1/3Mn1/3O2 Secondary Particles", Adv. Mater. (2016), DOI: 10.1002/adma.201601273.

