Electrodeposition (ED) of ZnO has been widely used to deposit transparent conducting films for optoelectronic applications. The film quality, and consequently the performance of the film, is highly dependent on both the nucleation and growth of ZnO. Current studies employ ex-situ experiments where the film is deposited, dried, and prepared for electron imaging. However, such techniques may introduce artefacts from partial dissolution of the film,1 adhesion of undissolved electrolytes, and drying effects. Hence a technique to investigate the nucleation and growth in situ is fundamental towards obtaining insights into the deposition behavior, thereby opening up possibilities for improved ZnO films for optoelectronic applications.
For in-situ x-ray imaging, a customized 3D-printed cell was designed and fabricated (Figure 1). Two ZnO morphologies, nanorods and nanoplates, were investigated by varying Zn2+ concentrations using deposition potentials of -0.75 V and -0.97 V (vs Ag/AgCl reference electrode) at 65 oC with continuous oxygen bubbling. Transmission x-ray microscopy (TXM) was conducted at Beam Line 6-2 with 2.0 s exposure at intervals of 2.2 s during electrodeposition. Absorption images with spatial resolutions of approximately 30 nm were acquired using a 9700 eV x-ray energy, which is directly above the Zn K-edge.
X-ray absorption images revealed that density of the nanostructures (both nanorods and nanoplates) did not exhibit a significant increase with increasing deposition time. This strongly suggests that an initial instantaneous nucleation occurs before growth takes place. For the nanorods, growth was accompanied by an increase in diameter. For the nanoplates, growth occurred with an increase in plate diameter with insignificant changes in plate thickness, which can be explained by an excess concentration of chloride ions that attenuates growth in the (002) orientation.2
At the less negative deposition potential of -0.75 V (vs Ag/AgCl), swirling blurred structures were observed up to 350 s after the start of the electrodeposition. These observations agree with the long-hypothesized mechanism, that ZnO precipitation occurs close to the working electrode surface before deposition onto the electrode and subsequent growth.3 Swirling was not observed at -0.97 V (vs Ag/AgCl reference electrode) due to the increased reaction rate for nitrate reduction to form hydroxyl ions at more negative potentials, which results in a rapid supersaturation and immediate nucleation. The ability to observe instantaneous versus delayed deposition highlights the advantage of using the TXM to gain insights into dynamic processes that occurs in nucleation and growth studies. The observations described in this paragraph can be observed in the supplementary video associated with the paper.
In this study, the direct nucleation and growth of electrodeposited ZnO nanorods and nanoplates in situ was thus successfully imaged. This approach removes artefacts that may arise from partial dissolution of ZnO, adhesion, and drying of the film arising from ex situ microscopy investigations. A long hypothesized ZnO electrodeposition mechanism was also verified through dynamic and direct observation. This study has the potential to be extended to other electrodeposition systems for both nucleation/growth and corrosion studies.
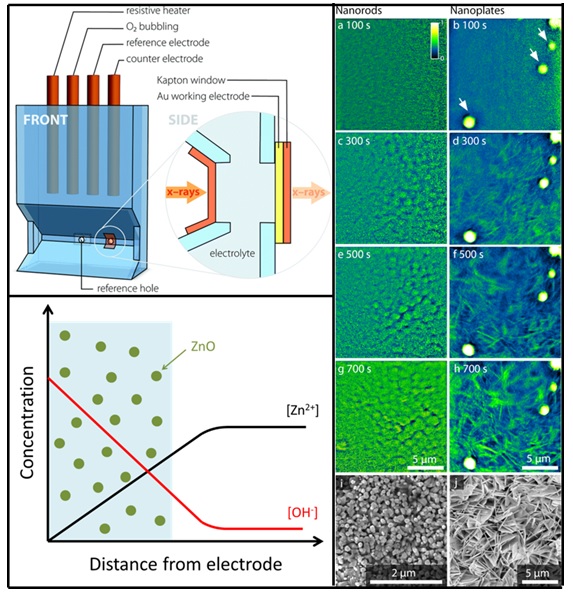
Figure 1. Schematic of customized 3D-printed electrochemical cell used (top-left figure). X-ray absorption images obtained at 100 s to 700 s of ZnO electrodeposition showing growth of ZnO nanorods (left column) and nanoplates (right column) with respective electron images (bottom-right figure). Diagrammatic representation of ZnO precipitation observed at less negative potential and long hypothesized in literature.
- D. M. Yebra, S. Kiil, C. E. Weinell and K. Dam-Johansen, "Dissolution Rate Measurements of Sea Water Soluble Pigments for Antifouling Paints: ZnO", Prog. Org. Coat. 56, 327 (2006).
- B. N. Illy, B. Ingham and M. P. Ryan, "Effect of Supersaturation on the Growth of Zinc Oxide Nanostructured Films by Electrochemical Deposition", Cryst. Growth Des. 10, 1189 (2010).
- T. Pauporté and D. Lincot, "Hydrogen Peroxide Oxygen Precursor for Zinc Oxide Electrodeposition II—Mechanistic Aspects", J. Electroanal. Chem. 517, 54 (2001).
S. E. R. Tay, A. E. Goode, J. Nelson Weker, A. A. Cruickshank, S. Heutz, A. E. Porter, M. P. Ryan and M. F. Toney, "Direct In situ Observation of ZnO Nucleation and Growth via Transmission X-ray Microscopy", Nanoscale 8, 1849 (2016), doi: 10.1039/c5nr07019h.

