Solar energy must be one of the primary energy sources as society transitions away from predominantly fossil fuels based economy. Currently, the overwhelming majority (>90%) of the photovoltaic (PV) market consists of silicon solar cells. While relatively inexpensive, this technology depends predominately on a screen-printed silver electrical front-contact and the formation of this contact uses a lead-based “frit”. Silver is expensive and lead is toxic. Thus, it is desirable to eliminate these materials in solar cells, and to achieve this, it is important to understand how the Ag-Si contact actually forms.
The silver front contact of the silicon solar cell is made by first screen-printing ink, containing the glass frit, silver, lead, and various additives. Once the ink is screen-printed in the desired pattern, the cell is then fired with a belt furnace that heats the silver ink to above 800°C at rates of up to 100°C/sec. During the firing the molten mixture etches through the silicon nitride (SiNx) antireflective-coating on the top of the solar cell and makes contact with the absorber layer. This process is crucial to the formation of a good electrical contact and an efficient solar cell. While the contact formation has been optimized with the “Edisonian approach”, the reaction sequence for the formation of the Ag-Si contact has not been fully understood. This understanding can help improve conventional contact materials, and can also be key to innovations that could reduce or replace the silver and lead in the inks, reducing costs and unwanted toxic metals.
To address this question, researchers at SSRL teamed with the National Renewable Energy Laboratory (NREL) to develop a chamber for use on SSRL Beam Line 7-2 that enables rapid thermal processing (RTP) at up to 1200°C at rates of 100°C/sec, while simultaneously collecting in-situ x-ray diffraction (XRD) in as little as 100 milliseconds. The RTP chamber was designed to simulate the belt furnace used for manufactured Ag-Si contacts by mimicking the belt furnace temperature profile. A schematic for this chamber is shown in Figure 1a [1]. With this new chamber the NREL-SSRL team could track the kinetics of the silver front contact formation, and reveal the reaction sequence that takes place during this process.
As reported in Nature Communications [2], during the firing process the frit melts above 500°C and wets the silver SiNx/ink interface. The lead oxide in the frit then reacts with the anti-reflective (SiNx) layer and forms metallic Pb via the reaction 2PbO + SiNx → 2Pb + SiO2 + ![]() N2. (Figure 1b), and the metallic Pb then alloys with the silver. Above 650°C, the silver dissolves into the molten frit and diffuses toward the emitter were the silver ions oxidize the Si via the reaction 2Ag2O + Si → SiO2 + 4Ag. Metallic silver is then deposited on the Si surface (forming the upside-down triangles of silver seen in Figure 1d and depicted in the schematic in Figure 1e). Upon cooling, the solubility of the silver in the molten frit decreases, which causes Ag nanocrystals to precipitate in the glass (Figure 1f); these are important for conductivity. Hence, to be effective, the process is required to produce an abundance of Ag in the glass, while avoiding any shunting that can be caused by excessive growth of the silver “spikes”. With the deeper understanding of the role silver and lead play in forming the silver front contact gained in this study, new innovations in front contact pastes are possible.
N2. (Figure 1b), and the metallic Pb then alloys with the silver. Above 650°C, the silver dissolves into the molten frit and diffuses toward the emitter were the silver ions oxidize the Si via the reaction 2Ag2O + Si → SiO2 + 4Ag. Metallic silver is then deposited on the Si surface (forming the upside-down triangles of silver seen in Figure 1d and depicted in the schematic in Figure 1e). Upon cooling, the solubility of the silver in the molten frit decreases, which causes Ag nanocrystals to precipitate in the glass (Figure 1f); these are important for conductivity. Hence, to be effective, the process is required to produce an abundance of Ag in the glass, while avoiding any shunting that can be caused by excessive growth of the silver “spikes”. With the deeper understanding of the role silver and lead play in forming the silver front contact gained in this study, new innovations in front contact pastes are possible.
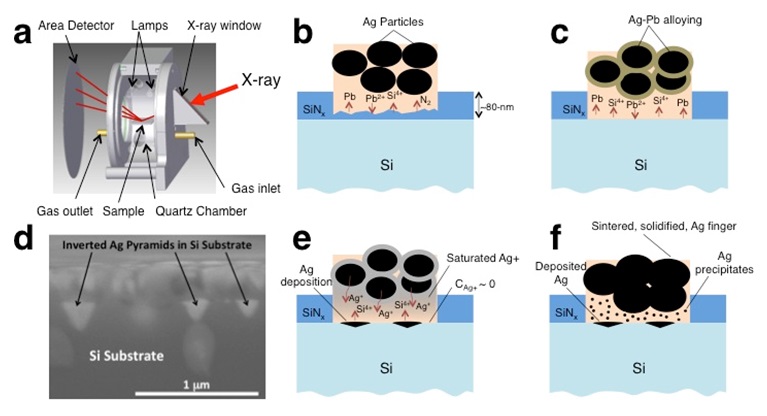
Figure 1 a) Schematic of RTP chamber; b) SiNx etching by PbO in frit; c) Ag-Pb alloying, d) SEM image at 100,000X magnification showing a cross-section of an Ag/frit/SiNx sample revealing inverted Ag pyramids penetrating the surface of the c-Si substrate; e) Ag+ transport through molten frit and deposition at Si surface, and f) resulting fired-contact morphology, with inclusion of Ag precipitates within the glass intermediate layer.
[1] Md. I. Ahmad et al., “Rapid Thermal Processing Chamber for In-situ X-ray Diffraction”, Rev. Sci. Instrum. 86, 013902 (2015).
[2] J. D. Fields, Md. I. Ahmad, V. L. Pool, J. Yu, D. G. Van Campen, P. A. Parilla, M. F. Toney and M. F. A. M. van Hest, "The Formation Mechanism for Printed Silver-contacts for Silicon Solar Cells", Nat. Commun. 7, 11143 (2016).
J. D. Fields, Md. I. Ahmad, V. L. Pool, J. Yu, D. G. Van Campen, P. A. Parilla, M. F. Toney and M. F. A. M. van Hest, "The Formation Mechanism for Printed Silver-contacts for Silicon Solar Cells", Nat. Commun. 7, 11143 (2016), DOI: 10.1038/ncomms11143.

