Managed aquifer recharge (MAR) is an increasingly used water enhancement strategy, which involves subsurface storage of water supplies in groundwater aquifers. While MAR projects have the potential to alleviate water deficits, they can also adversely impact groundwater quality by altering the native geochemistry of the aquifer and triggering the release of toxic contaminants native to aquifer sediments.
A particular contaminant of concern is arsenic, which is harmful to human and ecosystem health and ubiquitous in subsurface sediments around the world. Given a drinking water limit of 10 μg/L As,1 even extremely low concentration As pulses (on the order of a few μg/L) are enough to jeopardize groundwater quality and render numerous groundwater storage sites unusable. Unsafe concentrations of As release during MAR have been reported at numerous sites as reviewed in Neil et al.2 and can be attributed to various geochemical processes.
While numerous studies have focused on the fate of As in groundwater, in many cases, the pathways of As release are still not fully understood. Moreover, MAR projects can occur in a variety of different geochemical environments and can utilize a wide range of recharge water chemistries, meaning that there are numerous geochemical conditions that have not yet been evaluated. For example, the Groundwater Replenishment System (Orange County Water District, Orange County, CA) is an indirect potable reuse project in which wastewater is treated to potable standards. The high-purity, treated water is discharged into a series of infiltration basins to recharge local groundwater supplies. Generally, arsenic mobilization is not considered a problem within aerated systems due to the high affinity of soil and sediment minerals for the most oxidized form of arsenic, arsenate.3 However, in Orange County, CA, infiltration of recharge water to shallow aquifers has resulted in the release of As despite aerated conditions and low solid-phase concentrations of As.
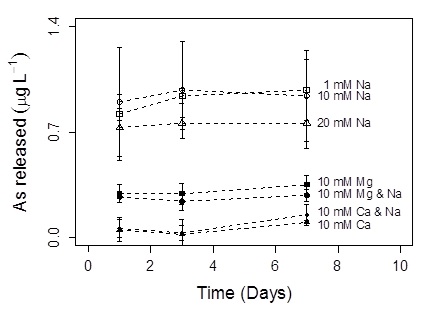
A series of laboratory experiments and spectroscopic analyses were used to elucidate the mechanism of As release from sediments underlying Orange County MAR basins. X-ray absorption spectroscopy conducted at SSRL Beam Line 11-2 revealed that As resides as arsenate, As(V), typically at more than 90%, and is located entirely in the clay fraction (<2 μm) of the sediments. This indicated that As release is controlled by interactions with clay minerals in the aquifer. Initial hypotheses focused on the high-purity nature of the recharge water which could strip the aquifer solids of their ionic strength, altering the surface charge of the clays and mobilizing As into the surrounding pore water. To test the effect of ionic strength, field sediments were incubated with recharge water modified with varying ionic strengths using Na+, Mg2+ and Ca2+ salts. Arsenic release from the sediments was monitored over time. Results indicate that when ionic strength is supplied as divalent cations, arsenic release is significantly suppressed compared to treatments in which ionic strength is supplied as a monovalent cation (Figure 1). Divalent cations possess a higher charge density and are a capable of forming cation bridging complexes between negatively charged clay particles and negatively charges aqueous arsenate species (H2AsO4- and HAsO42-).
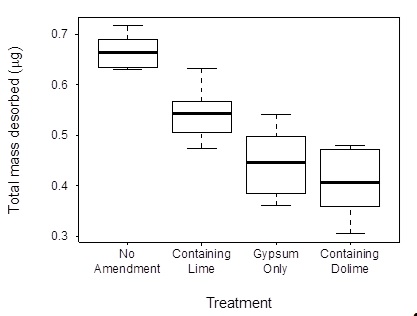
Figure 2: Box-and-whisker diagram comparing total mass of As eluted for various groundwater replenishment system (GWRS) amendments used in column experiments. “No amendment” refers to unmodified GWRS; “containing lime” and “containing dolime” refer to any treatments which included quicklime (Ca(OH)2) and dolomitic lime (CaO·MgO), respectively, while “gypsum only” refers to treatments of GWRS modified solely with gypsum (CaSO4·2H2O).
Divalent cations are commonly delivered during water treatment finishing steps as lime products. Accordingly, column experiments were conducted to analyze the release of As from field sediments under transport conditions and translate findings from the batch desorption experiments into potential water treatment options. Arsenic elution was monitored from continuous-flow column experiments using recharge water saturated with quicklime (Ca(OH)2), dolomitic lime (CaO·MgO), gypsum (CaSO4·2H2O) or combinations of the three. All divalent treatments decrease the release of As from sediments with dolomitic lime being the most effective treatment (Figure 2).
In summary, arsenic release from sediments underlying Orange County infiltration basins shows that MAR can mobilize As even in environments not traditionally considered vulnerable to As release. Laboratory studies reveal that As desorption can be corrected by targeting recharge water chemistries which minimize the propensity for As release. Specifically, the use of the common water amendments (lime and/or dolomitic lime) can provide recharge water with divalent cations and promote the formation of As cation bridging complexes within clay dominated sediments.
- US EPA. Arsenic Rule http://water.epa.gov/lawsregs/rulesregs/sdwa/arsenic/regulations.cfm (accessed Jan 12, 2015).
- C. W. Neil, Y. J. Yang and Y.-S. Jun, "Arsenic Mobilization and Attenuation by Mineral–water Interactions: Implications for Managed Aquifer Recharge", J. Environ. Monit. 14, 1772 (2012).
- P. L. Smedley and D. G. Kinniburgh, "A Review of the Source, Behaviour and Distribution of Arsenic in Natural Waters", Appl. Geochem. 17, 517 (2002).
S. Fakhreddine, J. Dittmar, D. Phipps, J. Dadakis and S. Fendorf, "Geochemical Triggers of Arsenic Mobilization during Managed Aquifer Recharge", Environ. Sci. Technol. 49, 7802 (2015), DOI: 10.1021/acs.est.5b01140.

