Improvements in the high-voltage cycling stability of lithium ion battery cathode materials are needed to enable the wide-spread adoption of renewable energy technologies such as electric vehicles. One cathode material which exhibits significant advantages over the commonly-used commercial material LiCoO2 in terms of higher capacity, increased thermal stability, and reduced cost is LiNi0.4Mn0.4Co0.2O2 (referred to as NMC). However, the substitution of Co by Mn and Ni results in a decrease in the electronic conductivity, leading to poor performance at high cycling rates, and increased reactivity with the electrolyte which results in a limited lifetime. To mitigate these effects, researchers from NREL have employed the use of single-walled carbon nanotubes in place of the typical polymer binder to increase rate-capability,[1] and an ultrathin Al2O3 coating prepared using atomic layer deposition to improve structural stability.[2] This approach enables remarkable high rate, highly stable performance at high voltages, representing an exciting development in Li-ion battery research.
An understanding of how the Al2O3 coating affords this structural stability is critical for further advances in the field. To this end, researchers from SSRL and NREL have taken a multimodal approach to thoroughly investigate the effect of the Al2O3 coating. Electrochemical impedance spectroscopy (EIS), operando synchrotron-based x-ray diffraction (XRD), and operando x-ray absorption near edge fine structure spectroscopy (XANES) have been utilized to characterize the structure and chemical evolution of the LiNi0.4Mn0.4Co0.2O2 cathode during cycling. Data were collected on both coated and uncoated electrodes using each technique during electrochemical cycling to high voltages, using typical coin cells for EIS measurements and coin cells modified to allow x-ray transmission for the XAS and XRD measurements.
From the EIS and XRD measurements, they were able to show that the Al2O3 coating prevents the severe reactions with the electrolyte seen in the bare electrodes at voltages > 4.4 V, without restricting the lithium intercalation/deintercalation. The operando XAS measurements revealed that the Al2O3 coating has an impact at much lower voltages, delaying the involvement of Co and Mn ions from the start of lithium deintercalation by protecting the surface chemistry. This protection promotes charge compensation via the oxidation of Ni by preventing the competing side reactions of the electrolyte with the highly active Ni oxide sites, thus enabling the observed high voltage, high rate cycling stability.
The knowledge gained here on the mechanism of the improved stability afforded by the Al2O3 coating will help to direct future developments to further improve the performance of this important class of material for Li-ion batteries. This study also highlights the power of a multimodal operando characterization approach applicable to the investigation of many different systems.
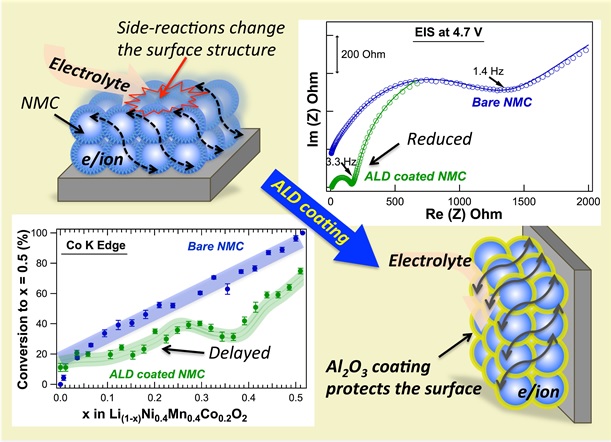
Figure 1. Illustration of the protection of the NMC electrode by the ALD Al2O3 coating with the electrochemical impedance spectroscopy data (top right) showing the prevention of the formation of a highly resistive surface layer at 4.7 V in the ALD coated electrode, with the delay in the conversion of the Co in the coated electrode compared to the bare as determined from the XANES data (bottom left).
- C. Ban, et al., "Extremely Durable High-Rate Capability of a LiNi0.4Mn0.4Co0.2O2 Cathode Enabled with Single-Walled Carbon Nanotubes", Adv. Eng. Mater. 1, 58 (2011).
- L. A. Riley, et al., "Electrochemical Effects of ALD Surface Modification on Combustion Synthesized LiNi 1/3Mn 1/3Co 1/3O2 as a Layered-cathode Material", J. Power Sources 196, 3317 (2011).
A. M. Wise, C. Ban, J. Nelson Weker, S. Misra, A. S. Cavanagh, Z. Wu, Z. Li, M. S. Whittingham, K. Xu, S. M. George and M. F. Toney, "Effect of Al2O3 Coating on Stabilizing LiNi0.4Mn0.4Co0.2O2 Cathodes", Chem. Mater. 27, 6146 (2015), DOI: 10.1021/acs.chemmater.5b02952.

