The human immune system relies on a network of protein sensors poised to detect foreign bacteria and viral pathogens. Researchers have discovered several families of receptors to explain how cells detect different types of microorganisms, but the evolutionary origin and specific signaling mechanism of these receptors often remain unclear. In a recent study, a team led by Philip Kranzusch (Howard Hughes Medical Institute and University of California-Berkeley), James Berger (Johns Hopkins University), and Jennifer Doudna (HHMI, UC-Berkeley and Lawrence Berkeley National Laboratory) has demonstrated that an enzyme encoded in the bacterial genome of Vibrio cholerae may point to an evolutionary connection between human innate immune signaling and bacterial pathogenesis.
A classic sign of pathogen replication is the presence of DNA in the cell cytosol. The human DNA genome is contained within the cell nucleus, and an aberrant localization of DNA outside of the nucleus is therefore recognized as a danger signal. The enzyme cyclic guanosine monophosphate (GMP)-adenosine monophosphate (AMP) synthase, or cGAS, is a cytosolic DNA sensor that upon activation produces a unique cyclic GMP-AMP dinucleotide named cGAMP (1). cGAS is the first human enzyme found to produce a cyclic dinucleotide signal, and unlike all previously discovered cyclic dinucleotides, cGAMP was shown to contain a unique 2ʹ–5ʹ linkage (2–4). After the initial characterization of cGAS/cGAMP by several groups, two important questions remained. If cGAS is the first discovered human cyclic dinucleotide synthase, where did the enzyme originate? Additionally, as the unique 2ʹ–5ʹ linkage is critical for optimal immune signaling, how does cGAS catalyze 2ʹ–5ʹ bond formation?
In order to provide insight into these questions, Kranzusch and Doudna envisioned that a new approach might be to first understand how bacterial cyclic dinucleotide synthases produce a related molecule. Cyclic dinucleotides were originally discovered as bacterial signaling molecules, and most bacteria encode cyclic di-AMP or cyclic di-GMP synthases. However, previous work had shown that an enzyme from Vibrio cholerae named DncV is a cyclic dinucleotide synthase that produces a 3ʹ–5ʹ linked cyclic GMP–AMP molecule (3ʹ–5ʹ cGAMP) similar to human cGAS (5). Although there is very little homology between human cGAS and Vibrio DncV (<10%), Kranzusch and Doudna reasoned that understanding how the bacterial enzyme synthesizes 3ʹ–5ʹ cGAMP would provide important clues to explain how human cGAS produces the unique 2ʹ–5ʹ cGAMP. The authors obtained crystals of Vibrio DncV and, with Beam Lines 11-1 and 12-2, as well as ALS Beam Line 8.3.1, were able to collect experimental phase information and determine the structures of DncV in a series of substrate-bound and catalytically trapped states.
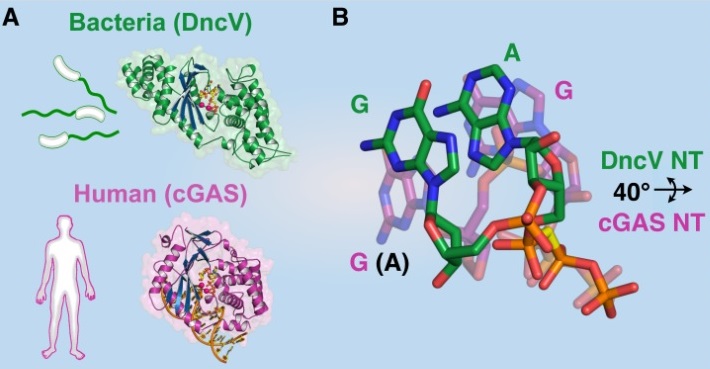
In spite of low sequence homology, the overall structure of DncV is very similar to that of human cGAS (6). The architectures of the catalytic active sites are nearly superimposable, and the structure of Vibrio DncV reveals that the bacterial enzyme is a closer homolog to human cGAS than to prokaryotic synthases (Figure 1A).
Using non-hydrolyzable nucleotide analogs, Kranzusch et al. were able to trap the enzyme and directly compare DncV and cGAS intermediate states (2). Although the active sites are structurally similar, DncV and cGAS reaction mechanisms proceed in opposing directions. Both enzymes synthesize cGAMP, but DncV positions guanosine triphosphate (GTP) as the first attacking nucleotide whereas cGAS first positions adenosine triphosphate (ATP) for attack.
Unexpected close structural homology between DncV and cGAS affords the rare opportunity to compare two closely related enzyme active sites that catalyze distinct chemical bonds. Superposing the two active sites reveals that the substrate nucleotides are rotated by approximately 40° between the two enzymes, where the cGAS substrate nucleotides are rotated downwards for 2ʹ–5ʹ cGAMP formation and the DncV substrate nucleotides are rotated upwards for 3ʹ–5ʹ cGAMP formation (Figure 1B). Alternative rotation of the substrate nucleotides can be explained by two positions of amino acid co-variation that contrast between the crystal structures. An extended glutamine side chain at the bottom of the pocket (site 1) and recessed isoleucine side chain at the top of the pocket (site 2) support the upward rotation of the bases in the DncV active site, while a recessed threonine side chain at site 1 below and an extended arginine side chain at site 2 above the bases induce the downward rotation of the bases in the cGAS active site. In this way, subtle variations in the active site geometry allow cGAS to specifically form 2ʹ–5ʹ cGAMP.
To prove that alternative extended and recessed amino acids control 2ʹ–5ʹ linkage specificity, the authors sought to reprogram cGAS to produce a new cGAMP species. Swapping the site 1 and site 2 cGAS positions with the corresponding DncV amino acids resulted in a cGAS variant that produced 3ʹ–5ʹ cGAMP. Mass-spectrometry and linkage-specific nuclease sensitivity were used to confirm 3ʹ–5ʹ cGAMP production. Importantly, the reprogrammed cGAS enzyme still responds to DNA and functions in cells within the innate immune signaling pathway.
Overall these results reveal a surprising evolutionary connection between a bacterial cyclic dinucleotide synthase and the recently discovered DNA sensor cGAS. Previously, evolutionary models had suggested that cGAS was derived from gene duplication of different metazoan innate immune receptors, but the study by Kranzusch et al. suggests that mammalian cGAS is evolutionarily related to ancestral prokaryotic enzymes. Additionally, the ability to reprogram the cGAS active site demonstrates enzymatic plasticity that allowed evolution of the unique 2ʹ–5ʹ cGAMP signaling molecule critical for human innate immunity.
- L. Sun, J. Wu, F. Du, X. Chen and Z. J. Chen, “Cyclic GMP-AMP Synthase is a Cytosolic DNA Sensor that Activates the Type I Interferon Pathway”, Science 339, 786 (2013).
- P. Gao, M. Ascano, Y. Wu, W. Barchet, Gaffney B.L. et al., “Cyclic [G(2ʹ,5ʹ)pA(3ʹ,5ʹ)p] is the Metazoan Second Messenger Produced by DNA-activated Cyclic GMP-AMP Synthase”, Cell 153, 1094 (2013).
- E. J. Diner, D. L. Burdette, S. C. Wilson, K. M. Monroe, C. A. Kellenberger C.A. et al., “The Innate Immune DNA Sensor cGAS Produces a Noncanonical Cyclic Dinucleotide that Activates Human STING”, Cell Rep. 3, 1355 (2013).
- A. Ablasser, M. Goldeck, T. Cavlar, T. Deimling, G. Witte G. et al., “cGAS Produces a 2ʹ–5ʹ-linked Cyclic Dinucleotide Second Messenger that Activates STING”, Nature 498, 380 (2013).
- B. W. Davies, R. W. Bogard, T. S. Young, J. J. Mekalanos, “Coordinated Regulation of Accessory Genetic Elements Produces Cyclic Dinucleotides for V. cholerae Virulence”, Cell 149, 358 (2012).
- P. J. Kranzusch, A. S. Y. Lee, J. M. Berger and J. A. Doudna, “Structure of Human cGAS Reveals a Conserved Family of Second-messenger Enzymes in Innate Immunity”, Cell Rep. 3, 1362 (2013).
P. J. Kranzusch, A. S. Y. Lee, S. C. Wilson, M. S. Solovykh, R. E. Vance, J. M. Berger and J. A. Doudna, "Structure-guided Reprogramming of Human cGAS Dinucleotide Linkage Specificity", Cell 158, 1011 (2014) doi: 10.1016/j.cell.2014.07.028

