Mercury is a well-known poison, but it is perhaps at its most dangerous when bound by organic groups to form organo mercury compounds (1). Such compounds are highly neurotoxic to mammals, but nevertheless have seen use as pesticides and also are made by microbial meabolism of mercuric ions. Several devastating mass-poisonings of human populations have been caused by organo mercury compounds. Organo mercury compounds exhibit an insidious latency in the development of toxic effects. Exposed humans may only develop toxic symptoms after a delay of several months. Adults are affected, but exposure in utero results in particularly severe consequences such as microcephaly, cerebropalsy, seizures, mental retardation, and other cruelly averse effects. Despite its toxicity and widespread occurance, many aspects of how organomercury compounds cause such deadly effects remains unknown.
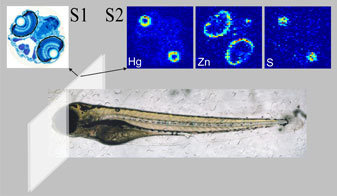
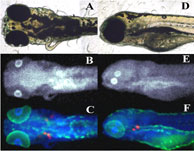
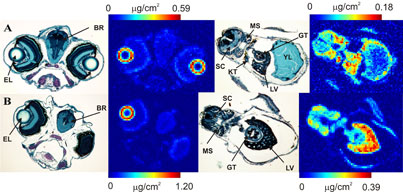
In an article published in the Proceedings of the National Academy of Sciences USA, Malgorzarta Korbas and co-workers have brought new insight into molecular mechanisms underlying organo mercury's toxicity. The team used X-ray fluorescence imaging conducted on SSRL beamlines 9-3 and 2-3 to study mercury accumulation in zebrafish (Danio rerio) larvae. Newly hatched zebrafish are relatively underdeveloped - for example, their eyes have yet to properly develop, and they still have a yolk sac attached - and they are therefore much used as a model organism for the study of vertebrate embryonic development and toxicology (2). Korbas and co-workers raised newly hatched zebrafish larvae in water containing low levels of methylmercury cysteinate (from 200 nm to 100 μM). The team first examined whole tricaine-anaesthetized larvae, using X-ray fluorescence imaging to obtain the distribution maps of mercury and other elements within the live fish. The researchers then investigated Hg distributions in sequential sections, with alternate sections being prepared for histology and X-ray fluorescence imaging (Figure 2). Strikingly, the greatest accumulation of methylmercury compounds was observed in the rapidly dividing layer of the lens epithelium, visible as rings at the periphery of the eye lenses (Figure 2,3). Lower levels of methylmercury were observed in brain, optic nerve and various other organs (Figure 3). The data suggest that the reported impairment of visual processes by mercury may arise not only from previously reported neurological effects, but also from direct effects on the ocular tissue.
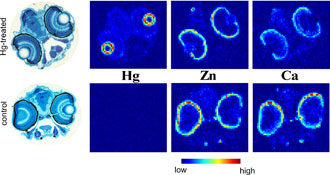
Control fish (Figure 4) showed no mercury signal, but displayed significantly higher calcium levels in the outer layers of the retina than the mercury exposed fish, suggesting that methylmercury might interfere with the natural calcium distribution as has previously been suggested (3).
This novel approach demonstrates synchrotron X-ray fluorescence imaging of zebrafish to be a powerful tool for investigating molecular toxicology of heavy metals. The method is equally applicable to the study of other elements of concern, such as arsenic, selenium, thallium and lead. The technique also provides an ideal tool for investigating drugs such as chelation agents (4) and can be applied to the study of essential metals and other elements of interest during normal development.
- Clarkson TW, Magos L (2006) The toxicology of mercury and its chemical compounds. Crit Rev Toxicol 36:609-662.
- Hill AJ, Teraoka H, Heideman W, Peterson RE (2005) Zebrafish as a model vertebrate for investigating chemical toxicity. Toxicol Sci 86:6-19.
- Gasso S, Cristofol RM, Selema G, Rosa R, Rodriguez-Farre E (2001) Antioxidant compounds and Ca2+ pathway blockers differentially protect against methylmercury and mercuric chloride neurotoxicity. J Neurosci Res 66:135-145.
- George GN, Prince RC, Gailer J, Buttigieg GA, Denton MB, Harris HH, Pickering IJ (2004) Mercury binding to the chelation therapy agents DMSA and DMPS, and the rational design of custom chelators for mercury. Chem Res Toxicol 17:999-1006.
Korbas M, Blechinger SR, Krone PH, Pickering IJ, George GN (2008) Localizing organomercury uptake and accumulation in zebrafish larvae at the tissue and cellular level. Proc Natl Acad Sci USA 105:12108-12112.

