Filamentous actin (F-actin), a biological rod-shaped protein, provides the structural framework in living cells. The assembly and organization of F-actin in vivo is controlled predominantly by actin binding proteins which locally crosslink actin into a rich variety of phases, including bundles and networks. Espins are one type of actin binding protein responsible for the formation of large parallel actin bundles. Espin is found in actin bundles in sensory cell microvilli, such as the stereocilia of cochlear hair cells. Within the cochlea of the inner ear, sound waves cause the basilar membrane to vibrate. These vibrations bend the stereocilia in the hair cells, which then trigger nerve impulses that are transmitted to the brain. Genetic mutations in espin's F-actin binding sites cause deafness. In this study crosslinked actin structures formed by mutated espins were studied in order to look into the potential link between the crosslinked bundle structure and deafness.
The structure of and interactions between the normal and mutant espin-actin complexes was systematically investigated using confocal microscopy and synchrotron small angle x-ray scattering (SAXS) performed at BL4-2 of the Stanford Synchrotron Radiation Laboratory and at the BESSRC-CAT (BL12-ID) at the Advanced Photon Source. The SAXS data shows how interactions between F-actin and different espin linkers are expressed in the system's self-assembled structure and phase behavior.
The mixtures of normal espin and F-actin showed well defined actin bundles in the confocal microscope images, which were found to be extremely well ordered hexagonal bundle of twisted actin filaments when studied via SAXS (Fig. 1). From the diffraction data we also measured the twisting of the helical F-actin filaments and found that it was increased by -0.9 degrees from the native helical twisting of F-actin. This suggests that the bundles of twisted filaments are under constant strain, which acts to maintain the stability of the bundle structure. From the large number of and narrow width of the hexagonally coordinated peaks we determined that these bundles were well very ordered and large. This corresponds to very rigid bundles; an important factor for bundles which are typically used as mechanical sensors (levers) in living cells.
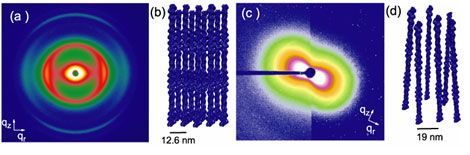
The structure of the bundles changes dramatically when normal espin is replaced with espin mutants that cause deafness. The deafness mutants have damaged actin binding sites and thus can be thought of as being less 'sticky' than the wild type espins. With these deafness mutations we could then assess the relationship between linker 'stickiness' and actin bundle formation. Damaging the actin binding site impairs the ability to form thick ordered bundles. The dominant deafness mutant (which has one good actin binding site, and one partial binding site) bundles actin at high espin concentrations creating a similar hexagonal structure to the normal espin-actin bundles but the inter-actin spacing was larger and more variable. At low espin concentrations, though, the hexagonal bundle peaks decreased in intensity and a new nematic-like peak appeared which displays a characteristic "bow-tie" pattern in 2D (Fig. 1). This indicates that the rods spontaneously oriented along an axis but they only had short-range positional ordering, not like the bundles which have long range ordering (Fig. 1). This nematic ordering is in addition to weak crosslinking which pulls the actin filaments close together, but not close enough to form bundles. A stronger mutant espin (which has only one actin binding site, the second being completely truncated by the mutation) was also studied, and for this recessive mutation, bundles never form, only the weakly crosslinked nematic phase is observed.
The observation of this crosslinked nematic-network phase was very exciting from a physics standpoint as new theories have been recently developed to explain physical relationship between the network and bundle phases of actin observed in cells, and the multitude of crosslinking proteins. By changing the preferred orientation of the crosslinking protein (does it like to form bundles or perpendicular networks) a rich phase behavior can predicted. However, the weakly crosslinked nematic phase observed with the deafness mutant espins, which are "weakly sticky" but still want to orient the actin filaments in bundles, has not yet been explored theoretically. This suggests that there is a new axis yet to be explored in the theoretical phase diagram of filaments and crosslinkers - the 'stickiness' axis.
The biological implications of a weakly crosslinked nematic phase in ear cell stereocilia show up predominantly in the bending stiffness. A weakly crosslinked nematic phase has a bending stiffness which is about a thousand times floppier, than a rigid bundle. This is in fact consistent with the observation that mutant espin cause malformed, floppy stereocilia. A thinner diameter bundle (which occurs even when the deafness mutant espins do bundle) also results in much floppier bundles. As a consequence the ear cannot respond to sound in the same way. When mixing mutant espin and normal espin (as is normally the case for the dominant mutant expression in humans), however, the bundling ability of the espin can be restored, and the bundles get slightly thicker. It is possible that this mechanism could be used to potentially 'rescue' this particular kind of pathology, particularly in the dominant mutant case, where partial expression of the normal espin is already possible. If gene expression could turn on the production of slightly more normal espin linkers, a kind of rescue attempt at restoring hearing could, in principle, be made.
Kirstin R. Purdy, James R. Bartles, and Gerard C. L. Wong; "Structural Polymorphism of the Actin-Espin System: A Prototypical System of Filaments and Linkers in Stereocilia", Phys. Rev. Lett. 98, 058105 (2007)

