Which came first, nucleic acids or proteins? This question is molecular biology's version of the "chicken-or-the-egg" riddle. Genes made of nucleic acids (DNA or RNA) contain the instructions for making proteins, but enzymes made of proteins are needed to replicate genes. For those who try to understand how life originated, this once seemed an intractable paradox.
The discovery 25 years ago that RNA can be enzymatic permits us to speculate that pre-biotic self-replicating molecules may have been RNAs (1,2). This is known as the "RNA World" hypothesis, and with the discovery of RNA catalysis, it is now possible to imagine a prebiotic 'RNA World' (or even one populated by early life forms) in which self-replicating ribozymes (RNA-based enzymes that possess the catalytic ability to copy themselves) accomplished both tasks, thus avoiding the potential "chicken-or-the-egg" conundrum (3).
But there's a catch.
In order to copy RNA, fragments or monomers that have 5'-triphosphates must be ligated together. This is true for modern polymerases, and is also the most likely mechanism by which a ribozyme self-replicase in an RNA World might function. Yet no one has found a modern natural ribozyme that catalyze this reaction (pictured right).
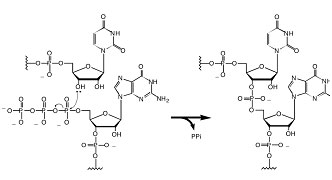
RNA in vitro evolution and selection has however enabled several research groups to discover RNA sequences that can in fact catalyze the required chemical reaction (shown above) for 5'-triphosphate RNA fragment ligation, and one group has even produced a primitive but functional RNA-based RNA polymerase ribozyme (4). This provides a Proof of Principle that RNA is capable of such feats, and absent time travel, this is likely the best we will be able to do.
As a first step to understanding what properties of ribozymes are of fundamental importance to a plausible RNA World evolutionary scenerio, and how RNA catalysts evolve in three-dimensional space, we have solved the structure of an RNA Ligase Ribozyme that catalyzes the assembly of RNA from a nucleotide-5'-triphosphate fragment.
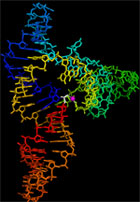
Dr. Michael Robertson in vitro selected a ligase ribozyme during his Ph.D. with Dr. Andrew Ellington that regioselectively catalyzes a 5'-3' RNA ligation reaction. He called this the L1 ligase. To better understand the details of how this ribozyme folds into a structure that permits it to catalyze this fundamental reaction, we have solved the structure of a catalytically active variant of his original ribozyme, which we call L1X6c (left).
The three-dimensional structure reveals tertiary contacts, including an invariant base triple, that help to position an essential Mg2+ at the ligation site, along with a potentially catalytically-relevant tightly-bound water molecule (right). Together with the RNA, these moieties help to guide proper regiospecific assembly of a 3' to 5' phosphodiester RNA backbone linkage. Specifically, the 2' and 3' protons of the ribose substrate have similar pKas, and formation of an improper 2' to 5' phosphodiester linkage would be equally probable to formation of a 3' to 5' linkage. The L1 ligase ribozyme appears to have evolved the capacity to bind a water molecule within hydrogen bonding distance to the 2'-oxygen, which might then lead to instant reprotonation of the aberrant nucleophile, should it become deprotonated. This would enable the the 3' to 5' reaction to be stereoselective in favor of the proper ligation product, i.e., the 3' to 5' phosphodiester linkage observed in all natural nucleic acids.
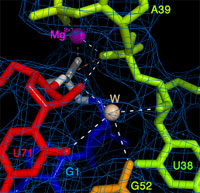
In summary, the L1 ligase ribozyme (as well as other in vitro evolved ligase ribozymes) is an empirical proof of principle that RNA is capable of catalyzing the phosphodiester isomerization reaction that would be required of any self-replicating RNA in a prebiotic RNA World. The three-dimensional structure gives us our first glimpse of how this reaction is catalyzed by the RNA in a regiospecific manner.
- K. Kruger, P. J. Grabowski, A. J. Zaug, J. Sands, D. E. Gottschling, and T. R. Cech. Self-splicing RNA: Autoexcision and autocyclization of the ribosomal RNA intervening sequence of tetrahymena. Cell, 1982, 31, 147-157.
- C. Guerrier-Takada, K. Gardiner, T. Marsh, N. Pace, and Sidney Altman. The RNA moiety of ribonuclease P is the catalytic subunit of the enzyme. Cell, 1983, 35, 849-857.
- R. F. Gesteland, J. F. Atkins, Eds., The RNA World. Cold Spring Harbor Laboratory Press, Plainview, New York, 1993.
- W. K. Johnston, P.J. Unrau, M.S. Lawrence, M.E. Glasner, and D.P. Bartel. 2001. RNA-catalyzed RNA- polymerization: Accurate and general RNA-templated primer extension. Science, 2001, 292, 1319-1325.
Michael P. Robertson and William G. Scott. The structural basis for ribozyme-catalyzed RNA assembly. Science, 2007, 315, 1549-1553.

