Allosteric Activation and Modulation of Pentameric Ligand-gated Ion Channels
April 2013 SSRL Science
Summary by Lori Ann White, SLAC Office of Communications
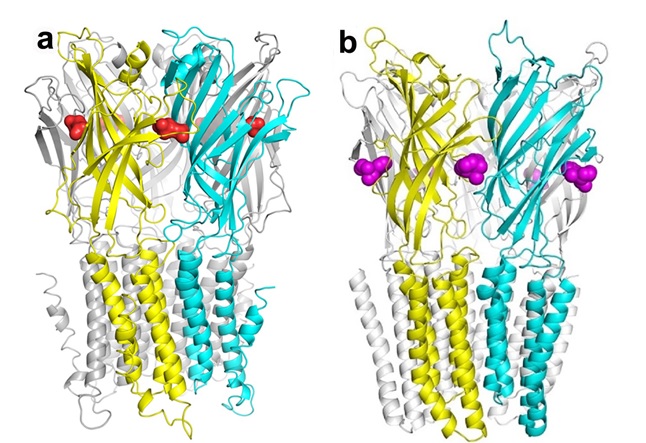
The structures studied by the researchers. (a) The 2.9-�-resolution structure of ELIC bound with ACh molecules (red). (b) The 2.99-�-resolution crystal structure of GLIC bound with five ketamine molecules (purple). |
A 2.9-� resolution structure of ELIC co-crystallized with acetylcholine (ACh) gave important clues regarding the pLGIC activation process. ACh-induced conformational rearrangements in ELIC's extra-cellular (EC) domain resembled those observed in some agonist-bound acetylcholine binding proteins. However, functional measurements and the crystal structure itself indicated that ACh acts as a competitive antagonist instead of an agonist for ELIC, bringing it to the verge of activation. Simply substituting -H for -CH3 in the ACh's choline group converted the ligand into an agonist. Because co-crystallization of ELIC with a high concentration of agonist is likely to produce crystallized proteins in a desensitized state, this high-resolution picture of ACh binding and the insights into the structural underpinning of agonism versus competitive antagonism are instrumental for designing new, optimized therapeutic drugs that can potentially suppress or enhance certain conformational states, thereby modulating the functions of Cys-loop receptors.
Cys-loop receptors are targets of general anesthetics, but pinpointing where and how the drugs act on these channels to modulate their functions remains a challenge. The researchers focused on the GLIC-ketamine complex. Although ketamine is commonly known as a dissociative anesthetic acting as a noncompetitive antagonist on the N-methyl-D-aspartate (NMDA) receptor, it is also a potent inhibitor of neuronal nAChRs, and the sites of action for ketamine at these receptors have not been identified previously. The study showed that ketamine inhibits GLIC at concentrations comparable to those that inhibit neuronal nAChRs. The crystal structure revealed a novel anesthetic binding site in a preexisting amphipathic cavity in GLIC's EC domain, and functional studies supported the importance of the site. The study provides compelling evidence for allosteric inhibition of pLGICs by anesthetics, as well as strongly suggesting that the anesthetic action of ketamine is based on more than just its action as an antagonist for the NMDA receptor.
Primary Citations
- J. Pan, Q. Chen, D. Willenbring, K. Yoshida, T. Tillman, O. B. Kashlan, A. Cohen, X.-P. Kong, Y Xu, and P. Tang, "Structure of ELIC Co-crystallized with Its Competitive Antagonist Acetylcholine", Nat. Commun. 3, 714 (2012) doi: 10.1038/ncomms1703
- J. Pan, Q. Chen, D. Willenbring, D. Mowrey, X.-P. Kong, A. Cohen, C. B. Divito, Y. Xu and P. Tang, "Structure of the Pentameric Ligand-Gated Ion Channel GLIC Bound with Anesthetic Ketamine", Structure 20, 1463 (2012) doi: 10.1016/j.str.2012.08.009
Related Links
- Science Highlight – HTML / PDF
- SSRL Science Highlights Archive
- SSRL Beam Lines
Contact
Pei Tang, University of Pittsburgh School of Medicine
