Nanoparticulate FeS as an Effective Redox Buffer to Prevent Uraninite (UO2) Oxidation
|
A major concern in the nuclear age is the contamination of soils and
groundwater with radionuclides from nuclear weapons and fuel production as well
as other human activities. One of the most prevalent contaminants is uranium
(U), whose mobility greatly depends on its oxidation state. Oxidized uranium,
U(VI), is soluble and can spread into groundwater. In contrast, reduced
uranium, U(IV), is often found in sparingly soluble phases such as solid
uraninite, UO2, and its low solubility reduces the environmental
risk. Naturally-occurring iron sulfide (FeS) is known to be an important
electron source for the reduction of uranium and oxidant scavenging.
Researchers from the University of Michigan and the Pacific Northwest National
Laboratory have recently used SSRL’s x-rays to shed light on the role of
FeS in protecting UO2 from reoxidation. The study, published in the
journal Geochimica et Cosmochimica Acta, may help evaluate the
long-term stability of uranium contaminations in the subsurface.
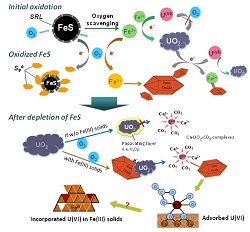
Simulating groundwater conditions, the scientists studied the oxidation of
UO2 nanoparticles by oxygen in the presence of nanoscale FeS in a
batch reactor. A time series of the reaction products were examined by X-ray
absorption spectroscopy (XAS) of the local uranium and sulfur environments
using SSRL’s Beam Lines 11-2 and 4-3, respectively. The team found that
the solid-phase uranium remained as U(IV)O2 while FeS rapidly
oxidized to elemental sulfur and Fe(III) oxyhydroxides.
Once all FeS had been oxidized, the researchers observed the subsequent
oxidative dissolution of UO2 into U(VI) species. The oxidized U(VI)
formed multinuclear carbonato complexes that adsorbed onto the Fe(III)
oxyhydroxide surfaces, indicating that adsorption is a major retention
mechanism for U(VI) species formed during UO2 oxidation. Thus, the
research reveals a dual role for FeS in immobilizing uranium in an
environmentally less harmful state: (1) FeS is an oxygen scavenger for
protecting UO2 against oxidation, and (2) its oxidation products are
adsorbents for U(VI) after FeS depletion.
Primary Citation
Y. Bi, S. P. Hyun, R. Kukkadapu and K. F. Hayes, "Oxidative Dissolution of UO2 in a Simulated Groundwater Containing Synthetic Nanocrystalline Mackinawite", Geochim. Cosmochim. Acta 102, 175 (2013) doi: 10.1016/j.gca.2012.10.032
Related Links
- Science Highlight – HTML / PDF
- SSRL Science Highlights Archive
- SSRL Beam Lines
Contact
Contacts: Yuqiang Bi and Kim F. Hayes, University of Michigan
