|
Ångström: One Ångström
is 10-10 meters, 1/10 of a nanometer. Atoms in molecules are
separated by about 1 Ångström. Since the wavelength of LCLS
x-rays are also in the range of 1 Ångström, the distribution
of x-rays scattered by an atom is influenced by the placement of its neighbor
atoms in a molecule. For this reason, x-ray scattering measurements provide
information on the structure of a molecule.
The wavelength of an x-ray is directly connected with the quantum of
energy that it can transfer to an atom if it is absorbed. The 1.5 - 15
ångström operating range of the LCLS corresponds to x-ray energies
from 8 keV to 800 eV. This means that LCLS x-rays can kick inner (K- and
L-) shell electrons out of most elements encountered in nature. Kicking
out electrons is the primary mechanism by which the LCLS delivers heat
to a sample.
 There are about as many Ångströms in a centimeter as there are
kilometers between the planet Venus and the sun.
There are about as many Ångströms in a centimeter as there are
kilometers between the planet Venus and the sun.
Femtosecond: One femtosecond is 10-15
second. A time this short is impossible to visualize in terms of
everyday experience, but it is a useful unit of time for describing atomic
and molecular dynamics. It takes about 10 femtoseconds for a hydrogen
atom to become attached to or detached from a molecule. Atoms heavier
than hydrogen are more massive and move more slowly, taking a few hundred
femtoseconds to enter or leave a binding site on a molecule. An electron
bound to an atom can transit from one atomic orbital to another in less
than a few femtoseconds. An atom or molecule in a gas or liquid will move
about one angstrom in a few femtoseconds.
 Light, which travels at 186,000 miles per second, moves no more than the
thickness of a sheet of paper in 150 femtoseconds.
Light, which travels at 186,000 miles per second, moves no more than the
thickness of a sheet of paper in 150 femtoseconds.
 There are about as many femtoseconds in a minute as there are minutes
in the entire history the universe.
There are about as many femtoseconds in a minute as there are minutes
in the entire history the universe.
Source:
Office of Basic Energy Science
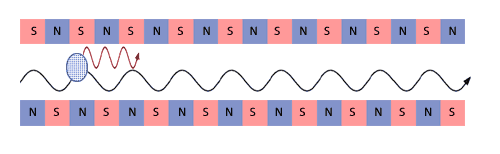
Self-Amplified Spontaneous Emission - SASE
An intense, highly collimated electron beam  travels
through an undulator magnet. The alternating north and south poles
of the magnet force the electron beam to travel on an approximately
sinusoidal trajectory, emitting synchrotron radiation travels
through an undulator magnet. The alternating north and south poles
of the magnet force the electron beam to travel on an approximately
sinusoidal trajectory, emitting synchrotron radiation  as
it goes. as
it goes. |
 |
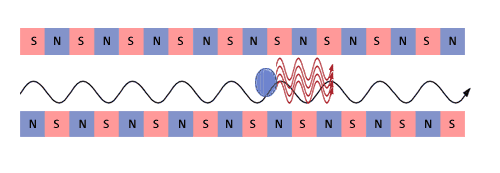
The electron beam and this synchrotron radiation travelling with it
are so intense that the electron motion is modified by the electromagnetic
fields of its own emitted synchrotron light. Under the influence of
both the undulator and its own synchrotron radiation, the electron
beam begins to form micro-bunches,  separated
by a distance equal to the wavelength of the emitted radiation. separated
by a distance equal to the wavelength of the emitted radiation. |
 |
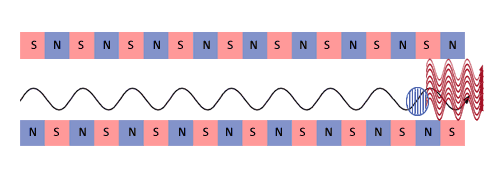
These micro-bunches begin to radiate as if they were single particles
with immense charge. The process reaches saturation when the micro-bunching
has gone as far as it can go. |
 |
|
 Glossary
Glossary Glossary
Glossary