Scientists Probe the Mechanism for Microbial Carbon Fixation
summary written by Raven Hanna
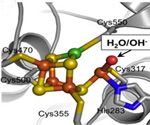
The smallest organisms should not be overlooked when finding solutions to the problem of increasing pollutants and greenhouse gases in our atmosphere. Each year, some microorganisms using the enzyme carbon monoxide dehydrogenase (CODH) take an estimated 100 million tons of carbon monoxide (CO) from our air, while others use CODH to produce 10 billion tons of acetate from carbon dioxide (CO2). CODH catalyzes the reversible reaction of CO to CO2, which different types of bacteria use in different chemical synthetic pathways. The enzyme has a complicated metal cluster, the C-cluster, which includes nickel, iron, and sulfur ions in its active site that performs this unusual chemistry. Understanding how this catalysis works may provide insights to chemical engineers wanting to duplicate this chemical process as a solution to the build-up of greenhouse gases.
A team of scientists led by Catherine Drennan of MIT used SSRL Beam Line 11-1 and a beam line at ALS to decipher the mechanism of CODH's reaction. They solved crystal structures which depict the C-cluster bound to a substrate and an inhibitor. Analyzing the structures, they were able to deduce the roles of the bound metals and the mechanism of the reaction.
This clearer understanding of how this microbial enzyme performs this difficult
chemistry may lead to methods to control pollutants and greenhouse gases in our
atmosphere. This work was published in Biochemistry.
To learn more about this research see the full Scientific Highlight
Kung, Y., Doukov, T.I., Seravalli, J., Ragsdale, S.W., Drennan, C.L. (2009) Crystallographic snapshots of cyanide- and water-bound C-clusters from bifunctional carbon monoxide dehydrogenase/acetyl-CoA synthase. Biochemistry. 48, 7432-7440.