
by David L. Clark, Los Alamos National Laboratory
A massive accelerated cleanup effort began in 1995. The key priority of site management and surrounding community leaders is the safe, accelerated closure of Rocky Flats. Kaiser-Hill and the DOE, working in close coordination with Rocky Flats stakeholders, are working on a plan to substantially complete the cleanup and closure of Rocky Flats by the aggressive goal of 2006. The closure of Rocky Flats is estimated to cost between $6 billion and $8 billion. [2]
The spatial distribution of plutonium in RFETS soils has been estimated,[3,4] with plutonium activities in surface soils ranging from 1,450 to 0.05 pCi/g, with the data showing a clear west-east trend away from an old drum storage site known as the 903 Pad. [5] More than 90% of the Pu is contained within the upper 10-12 cm of soils downwind of the 903 Pad (Fig 2).
 |
|
| Figure 1. From 1952 to 1989, Rocky Flats was the production site for nuclear and non-nuclear weapons components for the nation's nuclear weapons stockpile. | |
 |
|
| Figure 2. The 903 Pad was used in the 1950s to 1960s for storage, on bare ground, of more than 4,000 drums of plutonium-contaminated solvents and oils. The drums were removed in 1967 and 1968, and an asphalt pad was installed to control the spread of contamination. |
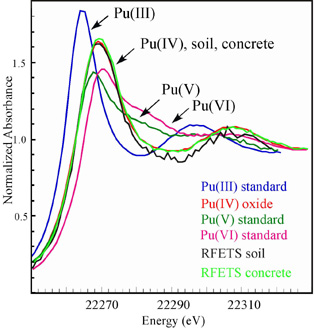
For plutonium studies, researchers prefer to use the LIII x-ray absorption edge (near 18,060 eV) because of its high absorption intensity.[8] However, in RFETS environmental samples there are other elements
 |
|
| Figure 3. Comparison of plutonium LII XANES spectra for plutonium in oxidation states III, IV, V, and VI with RFETS soil and concrete samples. |
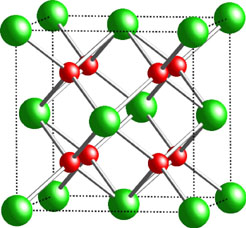
One of the soil samples was concentrated enough with respect to plutonium that XAFS data could be analyzed. XAFS curve fitting and data analysis revealed local structure features nearly identical to that for Pu in the solid PuO2 standard, the structure of which is illustrated in Fig 4, with one small exception. In addition to interatomic distances of 2.33 and 3.86 Å, the Fourier transforms from RFETS soil and concrete data show some small peaks at intermediate Pu-O distances between 2.3 and 3.0 Å which are consistent with additional Pu-OH or Pu-OH2 interactions which would be expected for hydrated PuO2 exposed to water in the environment. This was verified experimentally by examining the XAFS of a number of laboratory-prepared
 |
|
| Figure 4. Unit cell of fcc plutonium dioxide. Plutonium dioxide exhibits the well-known Fluorite crystal structure. Plutonium atoms are shown in green, and oxygen atoms are shown in red. This structure type gives rise to XAFS near-neighbor shells with Pu-O = 2.33, Pu-Pu = 2.38, and Pu-O = 4.52 Å, respectively. |
References:
- Draft Evaluation of Existing Data on Actinide Migration at the Rocky Flats Environmental Technology Site. 1996, RF/ER-96-0048.UN.
- see: www.refts.gov
- Krey, P. W.; Hardy, E. P. "Plutonium in Soil Around the Rocky Flats Plant", 1970, HASL-235, US AEC, Health and Safety Laboratory, New York.
- Litaor, M. I.; Thompson, M. L.; Barth, G. R.; Molzer, P. C., J. Environ. Qual., 1994, 23, 1231.
- DOE, 1995. Phase II RFI/RI Report for the 903 Pad, Mound, and East Trenches Area in Operable Unit No. 2, Volume 13, Appendix D (October, 1995)
- Clark, D. L., "The Chemical Complexities of Plutonium," Los Alamos Science, 2000, 26, 310
- Runde, W., "The Chemical Interactions of Actinides in the Environment," Los Alamos Science, 2000, 26, 338.
- Conradson, S. D.; Al-Mahamid, I.; Clark, D. L.; Hess, N. J.; Hudson, E. A.; Neu, M. P.; Palmer, P. D.; Runde, W. H.; Tait, C. D., "X-Ray Absorption Edges of Plutonium in Different Oxidation States," Polyhedron, 1998, 17, 599-602.
- Conradson, S. D., Appl. Spec. 1999, 52, 252.
- S. D. Conradson, D. L. Clark, M. P. Neu, W. Runde, C. D. Tait, "Characterizing the Plutonium Aquo Ions by XAFS Spectroscopy," Los Alamos Science, 2000, 26, 364.
- Hardy, E. P.; Krey, P. W., "Plutonium in the Environs of the Former Rocky Flats Plant," Health Phys. 1996, 71, 796.
SSRL Highlights Archive
