

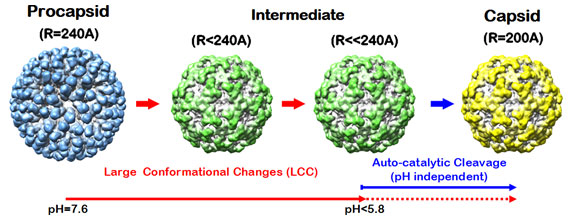
Most eukaryotic viruses undergo maturation when transitioning from the
noninfectious provirion to the infectious virion. Nudaurelia capensis omega
virus, NwV, is a T=4, non-enveloped, icosahedral,
single strand RNA virus, where T is the triangulation number defining an
icosahedral lattice of the virus capsid structure. Virus like particles (VLPs)
of NwV exhibit large pH-dependent conformational
changes (LCC) when the procapsid, purified at pH=7.6, (~480 Å) is exposed
to pH=5.0, resulting in ~400 Å particles (Figure 1). In response to the
LCC, an auto-proteolysis occurs in which each of 240 subunits is cleaved at
Asn570-Phe571 (1). We investigated this pH-induced
maturation by equilibrium and time-resolved small angle X-ray scattering (SAXS)
at SSRL beam line 4-2.
We showed that when the acidic interfaces of the NwV
subunits are protonated the electrostatic repulsion between adjacent subunits
is reduced, allowing the particle to undergo spontaneous size reduction through
a LCC. This condensation was studied at different pH values with equilibrium
SAXS allowing the determination of a titration curve that demonstrated a
continuous change in particle size with an overall particle pKa = ~5.8. An
N570T mutation of the NwV subunits, that does not
undergo the maturation cleavage, shows identical behavior to the wild type at
pH values between 7.6 and 6.0. However, they exhibit different maturation
properties at pH values between 5.8 and 5.0, with the mutant (uncleaved)
particle displaying systematically larger radii above pH 5.0.
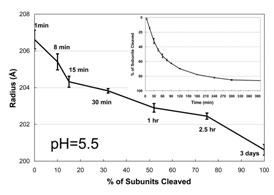
To directly determine the relationship between cleavage and particle size, slow
time resolved SAXS studies were carried out at pH 5.5 (the maximum pH at which
100% cleavage occurs in 3 days) (Figure 2). At pH=5.5, auto-proteolysis was
required to achieve the final mature size, whereas particles below pH 5.0 did
not require cleavage to achieve the final mature size.
The particle radius decreases from 236 Å to 207 Å in one minute and
then slowly decreases in size to ~200 Å as shown. The size change is
closely proportional
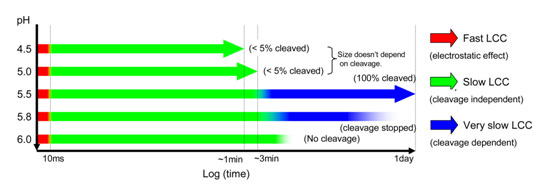
Finally, fast time resolved studies were performed at different pH values
(Figure 3). The maximum time resolution for the experiment is ~10 ms, and data
frames were recorded successively after the drop in pH using a stopped-flow
mixing apparatus. The results demonstrated 3 kinetic stages in the particle
condensation with each incremental drop in pH, the first at less than 10 ms,
the second in less than 5 seconds and the third in the 2-3 minute time regime
corresponding to the annealing described above. In addition to those stages,
the slow cleavage dependent stage (hours) is required at pH values between 5.8
and 5.5 (Figure 2). Those maturation events at the quaternary structure level
are reminiscent of protein folding, where there is rapid formation of a molten
globule, followed by different stages of polypeptide and side-chain annealing.
The study breaks new ground in understanding the energy landscape associated
with virus maturation, a process common in complex human viruses, and required
for a provirion to become an infectious virion. Encoded within the provirion
structure is a program that strengthens the capsid and activates an
auto-catalytic cleavage of the subunits.
Primary Citation
Matsui, T., Tsuruta, H. & Johnson, J.E. Balanced Electrostatic and Structural
Forces Guide the Large Conformational Change Associated with Maturation of T=4
Virus. Biophys. J., 98, 1337-1343.
References
to the fraction of subunits cleaved, emphasizing the role of cleavage at this
pH in the final stages of particle condensation. The kinetics of particle
cleavage at pH 5.5 is shown in the inset graph (1). The results indicated that
structural forces prior to cleavage counter the reduction in electrostatic
force and that the structural resistance due to the repulsive intersubunit
interaction is reduced when the auto-proteolysis occurs. These studies also
show that there is a significant period of protein annealing required for the
capsid to reach its equilibrium dimension. Based on the crystallographic
structure of the fully mature virion, we hypothesize that the annealing
involves positioning of the molecular switches associated with the T=4
quasi-symmetry as well as the formation of the active site for
auto-proteolysis.
SSRL is supported by the Department of Energy, Office of Basic Energy Sciences. The SSRL Structural Molecular Biology Program is supported by the Department of Energy, Office of Biological and Environmental Research, and by the National Institutes of Health, National Center for Research Resources, Biomedical Technology Program, and the National Institute of General Medical Sciences.