
 Since antiquity, magnetism has appeared to be a trick performed only by iron,
nickel, cobalt and a handful of rare alloys. But now the exclusive club of
magnetic elements officially has a new member: carbon. Using a proton beam and
advanced x-ray techniques, researchers at the Department of Energy's Stanford
Linear Accelerator Center (SLAC) in collaboration with colleagues from the
University of Leipzig and Lawrence Berkeley National Laboratory have finally
put to rest doubts about carbon's ability to be made magnetic. Although it has
long been suspected that carbon belongs on the short list of materials that can
be magnetic at room temperature, proof of that hypothesis has languished in
controversy for nearly a decade. Carbon's possible magnetic identity first
emerged when meteorites were found containing bits of the magnetized element,
but those flecks of carbon were packed in close proximity to nickel, leading to
the suspicion that the observed magnetism came from the nickel. Attempts to
prove that pure carbon can be magnetized have remained similarly unconvincing.
Now, a unique combination of proton accelerator and an x-ray microscope has
enabled researchers to dispel the last doubts about the existence of magnetic
carbon.
Since antiquity, magnetism has appeared to be a trick performed only by iron,
nickel, cobalt and a handful of rare alloys. But now the exclusive club of
magnetic elements officially has a new member: carbon. Using a proton beam and
advanced x-ray techniques, researchers at the Department of Energy's Stanford
Linear Accelerator Center (SLAC) in collaboration with colleagues from the
University of Leipzig and Lawrence Berkeley National Laboratory have finally
put to rest doubts about carbon's ability to be made magnetic. Although it has
long been suspected that carbon belongs on the short list of materials that can
be magnetic at room temperature, proof of that hypothesis has languished in
controversy for nearly a decade. Carbon's possible magnetic identity first
emerged when meteorites were found containing bits of the magnetized element,
but those flecks of carbon were packed in close proximity to nickel, leading to
the suspicion that the observed magnetism came from the nickel. Attempts to
prove that pure carbon can be magnetized have remained similarly unconvincing.
Now, a unique combination of proton accelerator and an x-ray microscope has
enabled researchers to dispel the last doubts about the existence of magnetic
carbon.
Ferromagnetism is an "ordering phenomena" in which the spins of neighboring
electrons are coupled together such that they point in the same direction. If
the temperature of the sample is elevated above a certain point, called
"Curie-temperature," however, the disorder caused by the thermal motion of the
atoms takes over and destroys the magnetic order. In fact, many different
materials show ferromagnetic behavior at low temperatures, below 5 Kelvin for
example, but only iron, cobalt, nickel and some alloys are useful ferromagnets
above room temperature and can be manufactured in large quantities. In recent
that looked like it might change as several groups reported suspected
ferromagnetic behavior in carbon, but the role of impurities in these samples
remained unclear. The key challenge of showing that a clean carbon sample can
exhibit ferromagnetism thus lingered. A particularly promising approach to
making carbon magnetic emerged from the group of Pablo Esquinazi at the
University Leipzig (Germany) in 2003. They irradiated clean carbon films with
an intense proton beam focused to a tiny spot of 2mm diameter. The proton
irradiation causes small distortions in the carbon lattice, which in turn cause
electron spins on neighboring atoms to align parallel. In a collaboration with
the Leipzig group we studied proton-irradiated samples at the Scanning
Transmission X-Ray Microscope (STXM) at the Berkeley Lab's Advanced Light
Source (ALS) to reveal the intrinsic carbon magnetism.
The STXM microscope is capable of addressing the magnetic properties of
different elements in a sample by using x-ray magnetic circular dichroism
(XMCD) in X-ray Absorption (XAS). In a STXM an incident x-ray beam is focused
on the sample by a lens called a "zoneplate" and the intensity of the
transmitted x-rays is measured on the detector. The sample is simultaneously
scanned perpendicular to the beam (Figure 1) ultimately yielding a full field
of view image. The absorption of x-rays is strongly enhanced when their energy
is chosen to match a core level resonance that appears when a core level
electron is excited into an empty valence state. These core-level resonances
appear at characteristic photon energies for different elements and one can
thus obtain information about the distribution of different elements in an
unknown sample. In addition to the elemental specificity, the transmission of
circular polarized x-rays at the resonance depends on the presence and
direction of a ferromagnetic moment (XMCD). It is therefore possible to obtain
information about the magnetism of the sample as well. Figure 2 shows the
results. A thin sample of carbon (t = 200 nm) is irradiated with a focused
protons beam leaving behind a magnetic ring. The images acquired using the STXM
at the carbon, iron, cobalt and nickel resonances reveal that the magnetic ring
only appears at the carbon resonance, and not at the others. It is noteworthy
that the detected magnetic signal is very small. Only the use of a modern
scanning transmission x-ray microscope at a state of the art x-ray source that
provides x-ray beams of high brilliance with variable polarization made it
possible to observe these tiny effects.
Figure 2: A carbon film is hit by a high-energy proton beam, causing the
magnetic moments of the atoms to align around the beam impact area and creating
a ring-shaped magnetic pattern that can be imaged with a magnetic-force
microscope (left). The x-ray microscope can then be used to "scan" the sample
for magnetism associated with other elements. The absence of a ring pattern in
scans for cobalt, nickel and iron prove that the sample contains only carbon
(right)
Harnessing the magnetic properties of carbon could one day revolutionize a
range of fields from nanotechnology to electronics. Carbon nanodevices could be
built one atom at a time, leading to miniaturized machines and lightweight
electronics. Magnetism, which forms the basis of information storage and
processing in computer hard drives, could be employed in novel ways in
tomorrow's electronic devices. The findings also underline the crucial
importance of modern x-ray science and instruments in basic research.
Stanford University operates SSRL for the Department of Energy's Office of
Science. The work at SSRL and ALS was supported by the U.S. Department of
Energy, Office of Basic Energy Sciences. The work in Leipzig was supported by
the German Research Foundation (DFG) and the European Union.
Primary Citation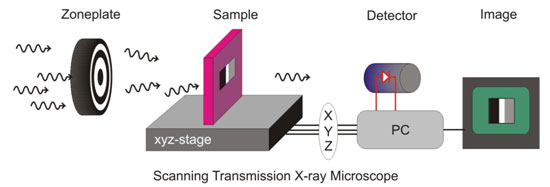
Figure 1: In a Scanning Transmission X-ray Microscope (STXM), x-rays are
focused onto a sample via a zoneplate. The sample can be moved perpendicular to
the x-ray beam, while the transmitted intensity is detected simultaneously to
produce a 2-D map of the x-ray absorption cross section of the sample using
computer software.

H. Ohldag, T. Tyliszczak, R. Höhne, D. Spemann, P. Equinazi, M. Ungureanu and
T. Butz, p-Electron Ferromagnetism in Metal-Free Carbon Probed by Soft X-ray
Dichroism", Phys. Rev. Lett., 98, 187204 (2007)