|
Related Links:
|
Thursday, 30 June 2005
More Oxidized than Rustsummary written by Heather Woods, SLAC Communication Office and Serena DeBeer George, SSRLNúria Aliaga-Alcalde,1 Serena DeBeer George,2 Bernd Mienert,1 Eckhard Bill,1 Karl Wieghardt1 and Frank Neese1
1Max Planck Institut für Bioanorganische Chemie, Mülheim an der
Ruhr, Germany
Iron metals oxidize to rust, losing electrons and gaining positive charge.
Iron metals typically exist in an oxidation state of +2 or +3 (2 or 3 electrons
less than a neutral iron atom). However, chemists have long thought that iron
compounds with even higher oxidation states play important roles in enabling
chemical reactions in metal-containing proteins. In recent years, scientists
have been able to synthesize and characterize numerous iron +4 compounds
[written Fe(IV)], but knew little about iron +5, Fe(V), compounds. Now
researchers, using SSRL, have characterized a genuine Fe(V) species, which is
even more oxidized and more positively charged than the iron in rust [Fe(III)]
or Fe(IV).
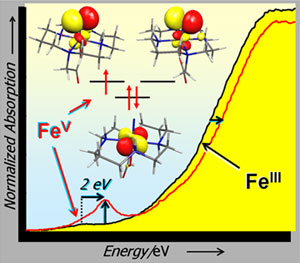
Frank Neese, Karl Wieghardt, and co-workers, including SSRL's Serena DeBeer
George, used X-ray absorption spectroscopy (XAS), combined with other
spectroscopic and computational results, to describe the compound. Tuned to be
sensitive to iron, XAS can pick up the amount of charge on the iron atom. The
XAS "K-edge" corresponds to the excitation of the most tightly bound electrons
in the iron atom. As the iron atom becomes more oxidized, the K-edge increases
in energy, providing a signature for Fe(V). This study represents the first
characterization of an Fe(V) species by XAS and serves as an important
experimental marker for characterization of other Fe(V) species.
|