
Todd W. Lane
1,
Mak A. Saito2, Graham N. George3, Ingrid J. Pickering
3, Roger C. Prince4 and François M.M. Morel5
1Biosystems Research Department, Sandia National Labs, Livermore, CA
2Marine Chemistry and Geochemistry Department, Woods Hole Oceanographic
Institution, Woods Hole, MA
3Department of Geological Sciences, University of Saskatchewan, Saskatoon,
Canada
4ExxonMobil Research and Engineering Company, Annandale, NJ
5Department of Geosciences, Princeton University, Princeton, NJ
Cadmium is known to be extremely toxic to mammals, and is generally viewed
alongside mercury an environmental problem and toxic element that is not used
by nature in any way. We have reported the characterization of a previously
unknown metalloenzyme from the marine diatom Thalassiosira weissflogii
that specifically uses cadmium to achieve its biological function. This work
shows that we need to revise our opinion of cadmium - it appears that it is not
only used biologically, but may play a vital role in the global carbon cycle.
Carbonic anhydrases catalyze the inter-conversion of carbonic acid and carbon
dioxide:
HCO3- + H+  CO2 +
H2O CO2 +
H2O
|
 |
|
|
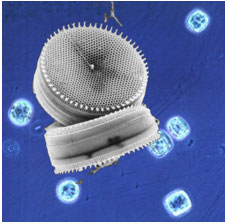
Figure 1. The marine diatom Thalassiosira weissflogii. The background shows an
optical micrograph, and the foreground shows a scanning electron micrograph of
the cells, which are disk-shaped, and ~10 µm across.
|
|
They are among the fastest enzymes known, with turnover numbers close to one
million per second. Photosynthesis in green plants can use only molecular
carbon dioxide, and not carbonic acid, and carbonic anhydrase thus represents
the first
step in the process of photosynthesis. In marine microalgae (e.g. diatoms),
carbonic acid is taken into the cell by a carbonic acid pump, converted to
CO2
by carbonic anhydrase, and then subsequently fed into the Calvin cycle by
ribulose bis-phosphate carboxylase, which uses molecular CO2.
All previously
characterized carbonic anhydrases incorporate an atom of zinc into the active
site, and these are divided into three categories - referred to as
a, b, g
carbonic anhydrases. The a-carbonic anhydrases are
by far the best studied, being found in animals (including mammals). They share
several highly conserved sequence elements, and contain zinc coordinated by
three histidines and (probably) a hydroxyl (1). The majority
of the higher plant isoforms make up the second class, and are referred to as
b-carbonic anhydrases. These contain no sequence
homology to the a-carbonic anhydrases, and contain
zinc ligated by two cysteine and one histidine, plus an activated water or
hydroxyl (2,3). A closely related coordination
is observed in the
b-carbonic anhydrases of the red alga
Porphyridium purpureum (4). The prototype
for the third (g) class has
 |
|
|
|
Figure 2.
Schematic diagram showing the role of carbonic anhydrase in the photosynthetic
uptake of carbon dioxide. | | been
reported for the bacterium Methanosarcina thermophila (5,6), and shows a zinc coordination resembling
the a-carbonic anhydrases. We have previously
reported that the diatom T. weissflogii when grown in the presence of
zinc produces zinc-containing carbonic anhydrase which has an active site
structure that is very similar to the a-carbonic
anhydrases, but with no sequence homology (7). We further proposed that this
should be designated as a fourth class the d-carbonic anhydrases, and concluded that the active site
structure was a striking example of convergent evolution at the molecular level
(7).
It is well established that the surface waters of the oceans, in which
 |
|
|
Figure 3.
Cadmium K-edge X-ray absorption spectra from purified enzyme (A) compared to
two tetrahedral thiolate-coordinated species -
[Cd(SPh)4](Me4N)2 (B, solid line),
Cd-phytochelatin (B, broken line), and the octahedral species
[Cd(H2O)6]2+ (C, solid line) and
[Cd(Imidazole)6](NO3)2 (C, broken line). The
similarity of spectra suggests the presence of non-octahedral site symmetry
possibly with cysteinyl ligands to the metal. |
microalgae such as diatoms flourish, are extremely low in zinc -
between 2 and 50 pico-molar. T. wiessflogii contains genes for two
discrete carbonic
anhydrases. This, together with the observation that adding cadmium allows the
diatom to grow (8), caused us to search for a specific cadmium
enzyme. Figure 3 shows the Cd K-edge spectra, collected on SSRL's beamline 7-3,
of the Cd carbonic anhydrase isolated from the diatom, and several different
model species. The concentration of the enzyme was only 7 µM, and the data
presented was the sum of 59 individual 25 minute scans. Comparison of the
near-edge spectra of Figure 3, allows us to formulate some conclusions about
the nature of the active site. The Cd-carbonic anhydrase spectrum clearly
resembles, but is not identical to, those of the tetrahedral models with a lot
of thiolate coordination. It seems very likely that the metal contains an
activated water or hydroxyl ligand, and a structure homologous to the higher
plant
b-carbonic
anhydrases seems plausible, but more definitive conclusions must await data
from more concentrated samples of the enzyme. The enzyme amino acid sequence is
distinct from all other carbonic anhydrases, and therefore represents yet
another discrete class, which we denote as
z-carbonic anhydrase.
Despite their microscopic size, marine phytoplankton are very numerous, and
make up a significant fraction of the world's plants. They are thus responsible
for a significant fraction of the cycling of atmospheric carbon dioxide through
photosynthesis. Cadmium is needed for this, at least in diatoms but probably in
other marine micro-algae too, so it may be that cadmium, rather than being an
environmentally detrimental element, is environmentally essential in the global
sense.
Primary Citation:
Lane, T.; Saito, M. A.; George, G. N.; Pickering, I. J.; Prince, R. C.; Morel,
F. F. M. "Isolation and Preliminary Characterization of a Cadmium Carbonic
Anhydrase from a Marine Diatom" Nature, 2005, 435, 42
References:
-
Fisher, Z.; Hernandez Prada, J. A.; Tu, C.; Duda, D.; Yoshioka, C.; An,
H.; Govindasamy, L.; Silverman, D. N.; McKenna, R. Biochemistry 2005,
44, 1097-1105 (and references therein).
-
Bracey, M. H.; Christiansen, J.; Tovar, P.; Cramer, S. P.; Bartlet, S.
G. Biochemistry, 1994, 33, 13126-13131.
-
Kimber, M. S.; Pai, E. F. EMBO J. 2000, 19, 1407-1418.
-
Mitsuhashi, S.; Mizushima, T.; Yamashita, E.; Yamamoto, M.; Kumasaka,
T.; Moriyama, H.; Ueki, T.; Miyachi, S.; Tsukihara, T. J. Biol.
Chem. 2000, 275, 5521-526.
-
Kisker, C.; Schindelin, H.; Alber, B. E.; Ferry, J. G.; Rees, D. C.
EMBO J. 1996, 15, 2323-2330.
-
Iverson, T. M.; Alber, B. E.; Kisker, C.; Ferry, J. G.; Rees, C. D.
Biochemistry 2000, 39, 9222-9231.
-
Cox, E. H.; McLendon, G. L.; Morel, F. M. M.; Prince, R. C.;
Pickering, I. J.; George G. N. Biochemistry 2000, 39, 12128-12130.
-
Morel F. F. M.; Price, N. M. Science, 2003, 300, 944-947 (and
references therein).
|
| PDF Version | | Lay Summary
|
| Highlights
Archive |
|
SSRL is supported by the Department of Energy, Office of Basic Energy Sciences.
The SSRL Structural Molecular Biology Program is supported by the Department of
Energy, Office of Biological and Environmental Research, and by the National
Institutes of Health, National Center for Research Resources, Biomedical
Technology Program, and the National Institute of General Medical Sciences.
|
|


 CO2 +
H2O
CO2 +
H2O


